ПЕРИОДИЧЕСКАЯ ТАБЛИЦА МЕНДЕЛЕЕВА
Еще в школе, сидя на уроках химии, все мы помним таблицу на стене класса или химической лаборатории. Эта таблица содержала классификацию всех известных человечеству химических элементов, тех фундаментальных компонентов, из которых состоит Земля и вся Вселенная. Тогда мы и подумать не могли, что таблица Менделеева бесспорно является одним из величайших научных открытий, который является фундаментом нашего современного знания о химии.
На первый взгляд, ее идея выглядит обманчиво просто: организовать химические элементы в порядке возрастания веса их атомов. Причем в большинстве случаев оказывается, что химические и физические свойства каждого элемента сходны с предыдущим ему в таблице элементом. Эта закономерность проявляется для всех элементов, кроме нескольких самых первых, просто потому что они не имеют перед собой элементов, сходных с ними по атомному весу. Именно благодаря открытию такого свойства мы можем поместить линейную последовательность элементов в таблицу, очень напоминающую настенный календарь, и таким образом объединить огромное количество видов химических элементов в четкой и связной форме. Разумеется, сегодня мы пользуемся понятием атомного числа (количества протонов) для того, чтобы упорядочить систему элементов. Это помогло решить так называемую техническую проблему «пары перестановок», однако не привело к кардинальному изменению вида периодической таблицы.
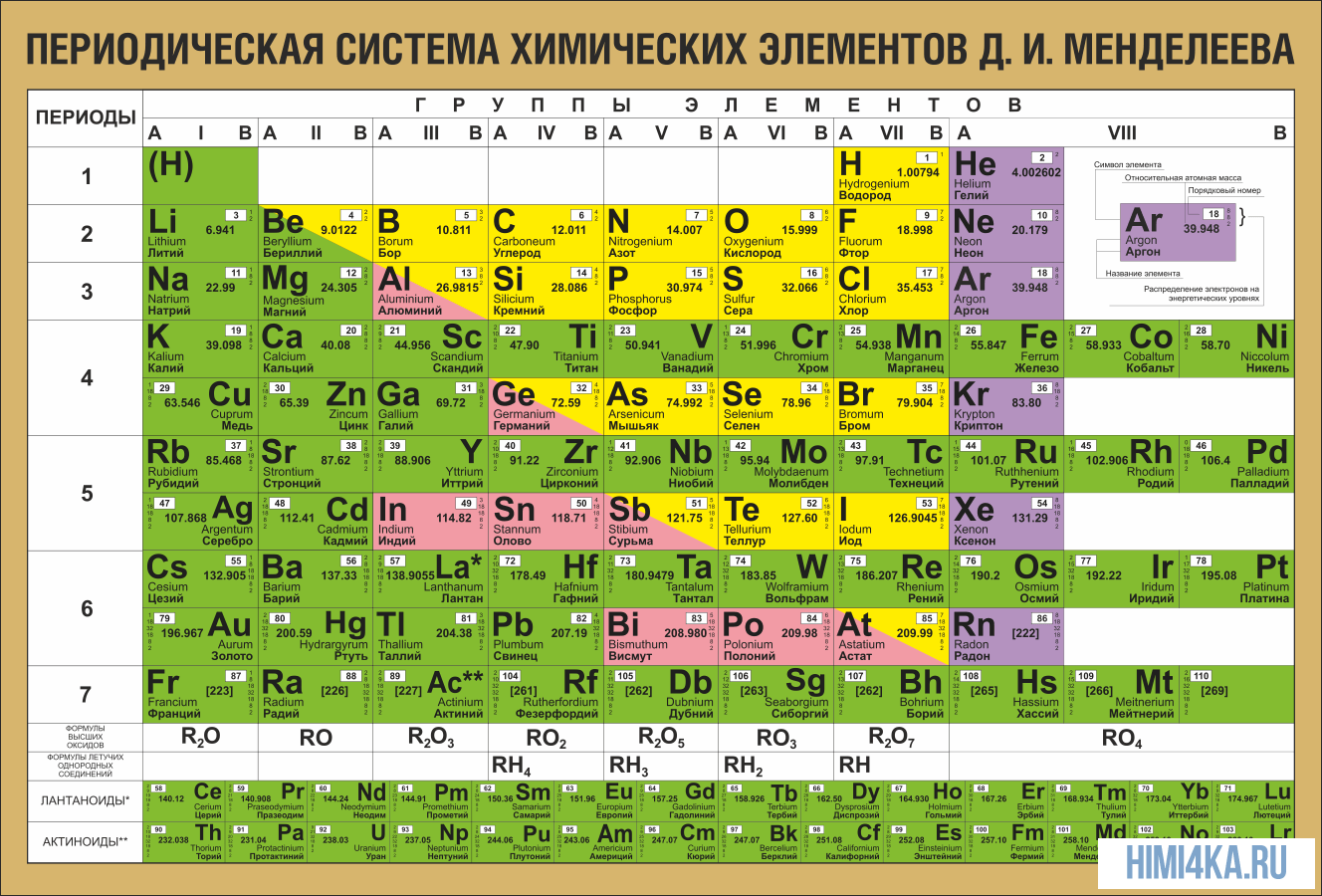
В периодической таблице Менделеева все элементы упорядочены с учетом их атомного числа, электронной конфигурации и повторяющихся химических свойств. Ряды в таблице называются периодами, а столбцы группами. В первой таблице, датируемой 1869 годом, содержалось всего 60 элементов, теперь же таблицу пришлось увеличить, чтобы поместить 118 элементов, известных нам сегодня.
Периодическая система Менделеева систематизирует не только элементы, но и самые разнообразные их свойства. Химику часто бывает достаточно иметь перед глазами Периодическую таблицу для того, чтобы правильно ответить на множество вопросов (не только экзаменационных, но и научных).
The YouTube ID of 1M7iKKVnPJE is invalid.
Периодический закон
Существуют две формулировки периодического закона химических элементов: классическая и современная.
Классическая, в изложении его первооткрывателя Д.И. Менделеева: свойства простых тел, а также формы и свойства соединений элементов находятся в периодической зависимости от величин атомных весов элементов.
Современная: свойства простых веществ, а также свойства и формы соединений элементов находятся в периодической зависимости от заряда ядра атомов элементов (порядкового номера).
Графическим изображением периодического закона является периодическая система элементов, которая представляет собой естественную классификацию химических элементов, основанную на закономерных изменениях свойств элементов от зарядов их атомов. Наиболее распространёнными изображениями периодической системы элементов Д.И. Менделеева являются короткая и длинная формы.
Группы и периоды Периодической системы
Группами называют вертикальные ряды в периодической системе. В группах элементы объединены по признаку высшей степени окисления в оксидах. Каждая группа состоит из главной и побочной подгрупп. Главные подгруппы включают в себя элементы малых периодов и одинаковые с ним по свойствам элементы больших периодов. Побочные подгруппы состоят только из элементов больших периодов. Химические свойства элементов главных и побочных подгрупп значительно различаются.
Периодом называют горизонтальный ряд элементов, расположенных в порядке возрастания порядковых (атомных) номеров. В периодической системе имеются семь периодов: первый, второй и третий периоды называют малыми, в них содержится соответственно 2, 8 и 8 элементов; остальные периоды называют большими: в четвёртом и пятом периодах расположены по 18 элементов, в шестом — 32, а в седьмом (пока незавершенном) — 31 элемент. Каждый период, кроме первого, начинается щелочным металлом, а заканчивается благородным газом.
Физический смысл порядкового номера химического элемента: число протонов в атомном ядре и число электронов, вращающихся вокруг атомного ядра, равны порядковому номеру элемента.
Свойства таблицы Менделеева
Напомним, что группами называют вертикальные ряды в периодической системе и химические свойства элементов главных и побочных подгрупп значительно различаются.
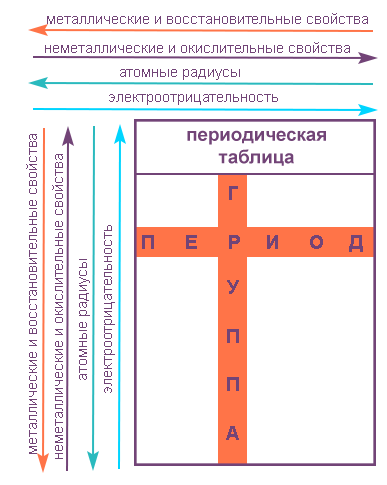
Свойства элементов в подгруппах закономерно изменяются сверху вниз:
- усиливаются металлические свойства и ослабевают неметаллические;
- возрастает атомный радиус;
- возрастает сила образованных элементом оснований и бескислородных кислот;
- электроотрицательность падает.
Все элементы, кроме гелия, неона и аргона, образуют кислородные соединения, существует всего восемь форм кислородных соединений. В периодической системе их часто изображают общими формулами, расположенными под каждой группой в порядке возрастания степени окисления элементов: R2O, RO, R2O3, RO2, R2O5, RO3, R2O7, RO4, где символом R обозначают элемент данной группы. Формулы высших оксидов относятся ко всем элементам группы, кроме исключительных случаев, когда элементы не проявляют степени окисления, равной номеру группы (например, фтор).
Оксиды состава R2O проявляют сильные основные свойства, причём их основность возрастает с увеличением порядкового номера, оксиды состава RO (за исключением BeO) проявляют основные свойства. Оксиды состава RO2, R2O5, RO3, R2O7 проявляют кислотные свойства, причём их кислотность возрастает с увеличением порядкового номера.
Элементы главных подгрупп, начиная с IV группы, образуют газообразные водородные соединения. Существуют четыре формы таких соединений. Их располагают под элементами главных подгрупп и изображают общими формулами в последовательности RH4, RH3, RH2, RH.
Соединения RH4 имеют нейтральный характер; RH3 — слабоосновный; RH2 — слабокислый; RH — сильнокислый характер.
Напомним, что периодом называют горизонтальный ряд элементов, расположенных в порядке возрастания порядковых (атомных) номеров.
В пределах периода с увеличением порядкового номера элемента:
- электроотрицательность возрастает;
- металлические свойства убывают, неметаллические возрастают;
- атомный радиус падает.
Элементы таблицы Менделеева
Щелочные и щелочноземельные элементы
К ним относятся элементы из первой и второй группы периодической таблицы. Щелочные металлы из первой группы — мягкие металлы, серебристого цвета, хорошо режутся ножом. Все они обладают одним-единственным электроном на внешней оболочке и прекрасно вступают в реакцию. Щелочноземельные металлы из второй группы также имеют серебристый оттенок. На внешнем уровне помещено по два электрона, и, соответственно, эти металлы менее охотно взаимодействуют с другими элементами. По сравнению со щелочными металлами, щелочноземельные металлы плавятся и кипят при более высоких температурах.
Показать / Скрыть текст
| Щелочные металлы | Щелочноземельные металлы |
| Литий Li 3 | Бериллий Be 4 |
| Натрий Na 11 | Магний Mg 12 |
| Калий K 19 | Кальций Ca 20 |
| Рубидий Rb 37 | Стронций Sr 38 |
| Цезий Cs 55 | Барий Ba 56 |
| Франций Fr 87 | Радий Ra 88 |
Лантаниды (редкоземельные элементы) и актиниды
Лантаниды — это группа элементов, изначально обнаруженных в редко встречающихся минералах; отсюда их название «редкоземельные» элементы. Впоследствии выяснилось, что данные элементы не столь редки, как думали вначале, и поэтому редкоземельным элементам было присвоено название лантаниды. Лантаниды и актиниды занимают два блока, которые расположены под основной таблицей элементов. Обе группы включают в себя металлы; все лантаниды (за исключением прометия) нерадиоактивны; актиниды, напротив, радиоактивны.
Показать / Скрыть текст
| Лантаниды | Актиниды |
| Лантан La 57 | Актиний Ac 89 |
| Церий Ce 58 | Торий Th 90 |
| Празеодимий Pr 59 | Протактиний Pa 91 |
| Неодимий Nd 60 | Уран U 92 |
| Прометий Pm 61 | Нептуний Np 93 |
| Самарий Sm 62 | Плутоний Pu 94 |
| Европий Eu 63 | Америций Am 95 |
| Гадолиний Gd 64 | Кюрий Cm 96 |
| Тербий Tb 65 | Берклий Bk 97 |
| Диспрозий Dy 66 | Калифорний Cf 98 |
| Гольмий Ho 67 | Эйнштейний Es 99 |
| Эрбий Er 68 | Фермий Fm 100 |
| Тулий Tm 69 | Менделевий Md 101 |
| Иттербий Yb 70 | Нобелий No 102 |
Галогены и благородные газы
Галогены и благородные газы объединены в группы 17 и 18 периодической таблицы. Галогены представляют собой неметаллические элементы, все они имеют семь электронов во внешней оболочке. В благородных газахвсе электроны находятся во внешней оболочке, таким образом с трудом участвуют в образовании соединений. Эти газы называют «благородными, потому что они редко вступают в реакцию с прочими элементами; т. е. ссылаются на представителей благородной касты, которые традиционно сторонились других людей в обществе.
Показать / Скрыть текст
| Галогены | Благородные газы |
| Фтор F 9 | Гелий He 2 |
| Хлор Cl 17 | Неон Ne 10 |
| Бром Br 35 | Аргон Ar 18 |
| Йод I 53 | Криптон Kr 36 |
| Астат At 85 | Ксенон Xe 54 |
| — | Радон Rn 86 |
Переходные металлы
Переходные металлы занимают группы 3—12 в периодической таблице. Большинство из них плотные, твердые, с хорошей электро- и теплопроводностью. Их валентные электроны (при помощи которых они соединяются с другими элементами) находятся в нескольких электронных оболочках.
Показать / Скрыть текст
| Переходные металлы |
| Скандий Sc 21 |
| Титан Ti 22 |
| Ванадий V 23 |
| Хром Cr 24 |
| Марганец Mn 25 |
| Железо Fe 26 |
| Кобальт Co 27 |
| Никель Ni 28 |
| Медь Cu 29 |
| Цинк Zn 30 |
| Иттрий Y 39 |
| Цирконий Zr 40 |
| Ниобий Nb 41 |
| Молибден Mo 42 |
| Технеций Tc 43 |
| Рутений Ru 44 |
| Родий Rh 45 |
| Палладий Pd 46 |
| Серебро Ag 47 |
| Кадмий Cd 48 |
| Лютеций Lu 71 |
| Гафний Hf 72 |
| Тантал Ta 73 |
| Вольфрам W 74 |
| Рений Re 75 |
| Осмий Os 76 |
| Иридий Ir 77 |
| Платина Pt 78 |
| Золото Au 79 |
| Ртуть Hg 80 |
| Лоуренсий Lr 103 |
| Резерфордий Rf 104 |
| Дубний Db 105 |
| Сиборгий Sg 106 |
| Борий Bh 107 |
| Хассий Hs 108 |
| Мейтнерий Mt 109 |
| Дармштадтий Ds 110 |
| Рентгений Rg 111 |
| Коперниций Cn 112 |
Металлоиды
Металлоиды занимают группы 13—16 периодической таблицы. Такие металлоиды, как бор, германий и кремний, являются полупроводниками и используются для изготовления компьютерных чипов и плат.
Показать / Скрыть текст
| Металлоиды |
| Бор B 5 |
| Кремний Si 14 |
| Германий Ge 32 |
| Мышьяк As 33 |
| Сурьма Sb 51 |
| Теллур Te 52 |
| Полоний Po 84 |
Постпереходными металлами
Элементы, называемые постпереходными металлами, относятся к группам 13—15 периодической таблицы. В отличие от металлов, они не имеют блеска, а имеют матовую окраску. В сравнении с переходными металлами постпереходные металлы более мягкие, имеют более низкую температуру плавления и кипения, более высокую электроотрицательность. Их валентные электроны, с помощью которых они присоединяют другие элементы, располагаются только на внешней электронной оболочке. Элементы группы постпереходных металлов имеют гораздо более высокую температуру кипения, чем металлоиды.
Показать / Скрыть текст
| Постпереходные металлы |
| Алюминий Al 13 |
| Галлий Ga 31 |
| Индий In 49 |
| Олово Sn 50 |
| Таллий Tl 81 |
| Свинец Pb 82 |
| Висмут Bi 83 |
Неметаллы
Из всех элементов, классифицируемых как неметаллы, водород относится к 1-й группе периодической таблицы, а остальные — к группам 13—18. Неметаллы не являются хорошими проводниками тепла и электричества. Обычно при комнатной температуре они пребывают в газообразном (водород или кислород) или твердом состоянии (углерод).
Показать / Скрыть текст
| Неметаллы |
| Водород H 1 |
| Углерод C 6 |
| Азот N 7 |
| Кислород O 8 |
| Фосфор P 15 |
| Сера S 16 |
| Селен Se 34 |
| Флеровий Fl 114 |
| Унунсептий Uus 117 |
А теперь закрепите полученные знания, посмотрев видео про таблицу Менделеева и не только.
Отлично, первый шаг на пути к знаниям сделан. Теперь вы более-менее ориентируетесь в таблице Менделеева и это вам очень даже пригодится, ведь Периодическая система Менделеева является фундаментом, на котором стоит эта удивительная наука.
Загрузить PDF
Загрузить PDF
Если таблица Менделеева кажется вам сложной для понимания, вы не одиноки! Хотя бывает непросто понять ее принципы, умение работать с ней поможет при изучении естественных наук. Для начала изучите структуру таблицы и то, какую информацию можно узнать из нее о каждом химическом элементе. Затем можно приступить к изучению свойств каждого элемента. Наконец, с помощью таблицы Менделеева можно определить число нейтронов в атоме того или иного химического элемента.
-
1
Таблица Менделеева, или периодическая система химических элементов, начинается в левом верхнем углу и заканчивается в конце последней строки таблицы (в нижнем правом углу). Элементы в таблице расположены слева направо в порядке возрастания их атомного номера. Атомный номер показывает, сколько протонов содержится в одном атоме. Кроме того, с увеличением атомного номера возрастает и атомная масса. Таким образом, по расположению того или иного элемента в таблице Менделеева можно определить его атомную массу.
- Атомная масса увеличивается слева направо и сверху вниз, поскольку в атомах элементов с бо́льшим номером содержится больше протонов и нейтронов. Каждый последующий элемент содержит больше протонов, чем предыдущий, поэтому возрастает и его атомная масса.
- В атомной массе не учитываются электроны, поскольку они весят намного меньше, чем протоны и нейтроны.[1]
-
2
Как видно, каждый следующий элемент содержит на один протон больше, чем предшествующий ему элемент. Это очевидно, если посмотреть на атомные номера. Атомные номера возрастают на один при движении слева направо. Поскольку элементы расположены по группам, некоторые ячейки таблицы остаются пустыми.[2]
- Например, первая строка таблицы содержит водород, который имеет атомный номер 1, и гелий с атомным номером 2. Однако они расположены на противоположных краях, так как принадлежат к разным группам.
-
3
Узнайте о группах, которые включают в себя элементы со схожими физическими и химическими свойствами. Элементы каждой группы располагаются в соответствующей вертикальной колонке. Как правило, они обозначаются одним цветом, что помогает определить элементы со схожими физическими и химическими свойствами и предсказать их поведение.[3]
Все элементы той или иной группы имеют одинаковое число электронов на внешней оболочке.[4]
- Водород можно отнести как к группе щелочных металлов, так и к группе галогенов. В некоторых таблицах его указывают в обеих группах.
- В большинстве случаев группы пронумерованы от 1 до 18, и номера ставятся вверху или внизу таблицы. Номера могут быть указаны римскими (например, IA) или арабскими (например,1A или 1) цифрами.
- При движении вдоль колонки сверху вниз говорят, что вы «просматриваете группу».
-
4
Узнайте, почему в таблице присутствуют пустые ячейки. Элементы упорядочены не только в соответствии с их атомным номером, но и по группам (элементы одной группы обладают схожими физическими и химическими свойствами). Благодаря этому можно легче понять, как ведет себя тот или иной элемент. Однако с ростом атомного номера не всегда находятся элементы, которые попадают в соответствующую группу, поэтому в таблице встречаются пустые ячейки.[5]
- Например, первые 3 строки имеют пустые ячейки, поскольку переходные металлы встречаются лишь с атомного номера 21.
- Элементы с атомными номерами с 57 по 71 относятся к редкоземельным элементам, и обычно их выносят в отдельную подгруппу в нижнем правом углу таблицы.
-
5
Каждая строка таблицы представляет собой период. Все элементы одного периода имеют одинаковое число атомных орбиталей, на которых расположены электроны в атомах. Количество орбиталей соответствует номеру периода. Таблица содержит 7 строк, то есть 7 периодов.[6]
- Например, атомы элементов первого периода имеют одну орбиталь, а атомы элементов седьмого периода — 7 орбиталей.
- Как правило, периоды обозначаются цифрами от 1 до 7 слева таблицы.
- При движении вдоль строки слева направо говорят, что вы «просматриваете период».
-
6
Научитесь различать металлы, металлоиды и неметаллы. Вы лучше будете понимать свойства того или иного элемента, если сможете определить, к какому типу он относится. Для удобства в большинстве таблиц металлы, металлоиды и неметаллы обозначаются разными цветами. Металлы находятся в левой, а неметаллы — в правой части таблицы. Металлоиды расположены между ними.[7]
- Помните, что по свойствам водород можно отнести как к щелочным металлам, так и к галогенам, поэтому он может иметь разный цвет и располагаться в двух ячейках.
- Металлические элементы блестят, являются твердыми при комнатной температуре (за исключением ртути), хорошо проводят тепло и электрический заряд и достаточно пластичны.
- Неметаллы не имеют выраженного блеска, плохо проводят тепло и электрический заряд, обладают низкой пластичностью. Как правило, при комнатной температуре неметаллические элементы представляют собой газ, но при определенных температурах и давлениях они могут принимать жидкое или твердое состояние.
- Металлоиды совмещают в себе свойства металлов и неметаллов.[8]
Реклама
-
1
Каждый элемент обозначается одной или двумя латинскими буквами. Как правило, символ элемента приведен крупными буквами в центре соответствующей ячейки. Символ представляет собой сокращенное название элемента, которое совпадает в большинстве языков. При проведении экспериментов и работе с химическими уравнениями обычно используются символы элементов, поэтому полезно помнить их.[9]
- Обычно символы элементов являются сокращением их латинского названия, хотя для некоторых, особенно недавно открытых элементов, они получены из общепринятого названия. К примеру, гелий обозначается символом He, что близко к общепринятому названию в большинстве языков. В то же время железо обозначается как Fe, что является сокращением его латинского названия.
-
2
Обратите внимание на полное название элемента, если оно приведено в таблице. Это «имя» элемента используется в обычных текстах. Например, «гелий» и «углерод» являются названиями элементов. Обычно, хотя и не всегда, полные названия элементов указываются под их химическим символом.[10]
- Иногда в таблице не указываются названия элементов и приводятся лишь их химические символы.
-
3
Найдите атомный номер. Обычно атомный номер элемента расположен вверху соответствующей ячейки, посередине или в углу. Он может также находиться под символом или названием элемента. Элементы имеют атомные номера от 1 до 118.[11]
- Атомный номер всегда является целым числом.
-
4
Помните о том, что атомный номер соответствует числу протонов в атоме. Все атомы того или иного элемента содержат одинаковое количество протонов. В отличие от электронов, количество протонов в атомах элемента остается постоянным. В противном случае получился бы другой химический элемент![12]
- По атомному номеру элемента можно также определить количество электронов и нейтронов в атоме.
-
5
Обычно количество электронов равно числу протонов. Исключением является тот случай, когда атом ионизирован. Протоны имеют положительный, а электроны — отрицательный заряд. Поскольку атомы обычно нейтральны, они содержат одинаковое количество электронов и протонов. Тем не менее, атом может захватывать электроны или терять их, и в этом случае он ионизируется.[13]
- Ионы имеют электрический заряд. Если в ионе больше протонов, то он обладает положительным зарядом, и в этом случае после символа элемента ставится знак «плюс». Если ион содержит больше электронов, он имеет отрицательный заряд, что обозначается знаком «минус».
- Знаки «плюс» и «минус» не ставятся, если атом не является ионом.
Реклама
-
1
Найдите атомную массу. Обычно атомную массу указывают внизу ячейки, под символом элемента. Атомная масса представляет собой сумму масс частиц, составляющих ядро атома, то есть протонов и нейтронов. Однако элементы имеют изотопы, поэтому атомная масса является средней величиной с учетом их распространенности в природе.[14]
- Поскольку атомная масса представляет собой среднюю величину, для большинства элементов она записывается в виде десятичной дроби.
- Хотя может показаться, что атомная масса всегда возрастает при движении слева направо и сверху вниз, это справедливо не во всех случаях.
-
2
Найдите массовое число конкретного элемента. Поскольку в атомной массе учитываются все изотопы элемента, для определения массового числа достаточно округлить приведенную в таблице атомную массу до ближайшего целого числа.[15]
- Например, атомная масса углерода составляет 12,011, после округления получаем 12. Для железа, атомная масса которого равна 55,847, получаем 56.
-
3
Вычтите из массового числа атома его атомный номер, чтобы найти количество нейтронов. Масса атома представляет сумму масс протонов и нейтронов. Ввиду этого можно легко определить число нейтронов — достаточно вычесть из массового числа атомный номер, то есть количество протонов![16]
- Используйте следующую формулу: Число нейтронов = Массовое число – Число протонов.
- Например, массовое число углерода равно 12, и атом углерода имеет 6 протонов. Поскольку 12 – 6 = 6, атом углерода содержит 6 нейтронов.
- Массовое число железа составляет 56, и его атом содержит 26 протонов. Так как 56 – 26 = 30, в атоме железа содержится 30 нейтронов.
- Изотопы элементов имеют разное количество нейтронов, благодаря чему меняется их атомная масса.
Реклама
Советы
- Многим сложно освоить таблицу Менделеева. Не расстраивайтесь, если столкнетесь с определенными трудностями!
- Хотя цвета могут меняться, они передают одну и ту же информацию.
- Встречаются упрощенные таблицы. Например, в таблице могут содержаться лишь символы элементов и их атомные номера. Выберите такую таблицу, которая подходит для ваших целей.
Реклама
Об этой статье
Эту страницу просматривали 250 557 раз.
Была ли эта статья полезной?
Как пользоваться таблицей Менделеева? Для непосвященного человека читать таблицу Менделеева – всё равно, что для гнома смотреть на древние руны эльфов. А таблица Менделеева может рассказать о мире очень многое.
Помимо того, что сослужит вам службу на экзамене, она еще и просто незаменима при решении огромного количества химических и физических задач. Но как ее читать? К счастью, сегодня этому искусству может научиться каждый. В этой статье расскажем, как понять таблицу Менделеева.
Периодическая система химических элементов (таблица Менделеева) – это классификация химических элементов, которая устанавливает зависимость различных свойств элементов от заряда атомного ядра.
История создания Таблицы
Дмитрий Иванович Менделеев был не простым химиком, если кто-то так думает. Это был химик, физик, геолог, метролог, эколог, экономист, нефтяник, воздухоплаватель, приборостроитель и педагог. За свою жизнь ученый успел провести фундаментально много исследований в самых разных областях знаний. Например, широко распространено мнение, что именно Менделеев вычислил идеальную крепость водки – 40 градусов.
Не знаем, как Менделеев относился к водке, но точно известно, что его диссертация на тему «Рассуждение о соединении спирта с водой» не имела к водке никакого отношения и рассматривала концентрации спирта от 70 градусов. При всех заслугах ученого, открытие периодического закона химических элементов – одного их фундаментальных законов природы, принесло ему самую широкую известность.
Существует легенда, согласно которой периодическая система приснилась ученому, после чего ему осталось лишь доработать явившуюся идею. Но, если бы все было так просто.. Данная версия о создании таблицы Менделеева, по-видимому, не более чем легенда. На вопрос о том, как была открыта таблица, сам Дмитрий Иванович отвечал: «Я над ней, может быть, двадцать лет думал, а вы думаете: сидел и вдруг… готово»
В середине девятнадцатого века попытки упорядочить известные химические элементы (известно было 63 элемента) параллельно предпринимались несколькими учеными. Например, в 1862 году Александр Эмиль Шанкуртуа разместил элементы вдоль винтовой линии и отметил циклическое повторение химических свойств.
Химик и музыкант Джон Александр Ньюлендс предложил свой вариант периодической таблицы в 1866 году. Интересен тот факт, что в расположении элементов ученый пытался обнаружить некую мистическую музыкальную гармонию. В числе прочих попыток была и попытка Менделеева, которая увенчалась успехом.
В 1869 году была опубликована первая схема таблицы, а день 1 марта 1869 года считается днем открытия периодического закона. Суть открытия Менделеева состояла в том, что свойства элементов с ростом атомной массы изменяются не монотонно, а периодически.
Первый вариант таблицы содержал всего 63 элемента, но Менделеев предпринял ряд очень нестандартных решений. Так, он догадался оставлять в таблице место для еще неоткрытых элементов, а также изменил атомные массы некоторых элементов. Принципиальная правильность закона, выведенного Менделеевым, подтвердилась очень скоро, после открытия галлия, скандия и германия, существование которых было предсказано ученым.
Современный вид таблицы Менделеева
Ниже приведем саму таблицу
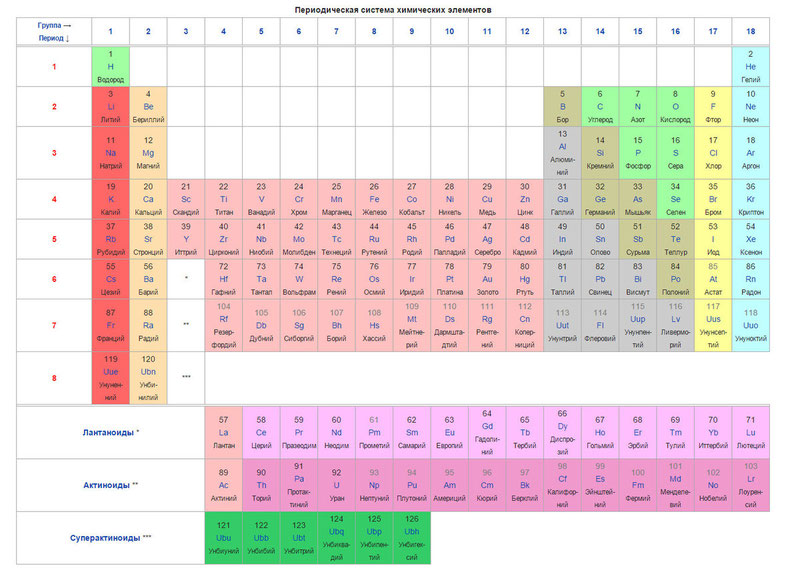
Сегодня для упорядочения элементов вместо атомного веса (атомной массы) используется понятие атомного числа (числа протонов в ядре). В таблице содержится 120 элементов, которые расположены слева направо в порядке возрастания атомного числа (числа протонов)
Столбцы таблицы представляют собой так называемые группы, а строки – периоды. В таблице 18 групп и 8 периодов.
- Металлические свойства элементов при движении вдоль периода слева направо уменьшаются, а в обратном направлении – увеличиваются.
- Размеры атомов при перемещении слева направо вдоль периодов уменьшаются.
- При движении сверху вниз по группе увеличиваются восстановительные металлические свойства.
- Окислительные и неметаллические свойства при движении вдоль периода слева направо увеличиваются.
Что мы узнаем об элементе по таблице? Для примера, возьмем третий элемент в таблице – литий, и рассмотрим его подробно.
Первым делом мы видим сам символ элемента и его название под ним. В верхнем левом углу находится атомный номер элемента, в порядке которого элемент расположен в таблице. Атомный номер, как уже было сказано, равен числу протонов в ядре. Число положительных протонов, как правило, равно числу отрицательных электронов в атоме (за исключением изотопов).
Атомная масса указана под атомным числом (в данном варианте таблицы). Если округлить атомную массу до ближайшего целого, мы получим так называемое массовое число. Разность массового числа и атомного числа дает количество нейтронов в ядре. Так, число нейтронов в ядре гелия равно двум, а у лития – четырем.
Вот и закончился наш курс «Таблица Менделеева для чайников». В завершение, предлагаем вам посмотреть тематическое видео, и надеемся, что вопрос о том, как пользоваться периодической таблицей Менделеева, стал вам более понятен. Напоминаем, что изучать новый предмет всегда эффективнее не одному, а при помощи опытного наставника. Именно поэтому, никогда не стоит забывать о студенческом сервисе, который с радостью поделится с вами своими знаниями и опытом.
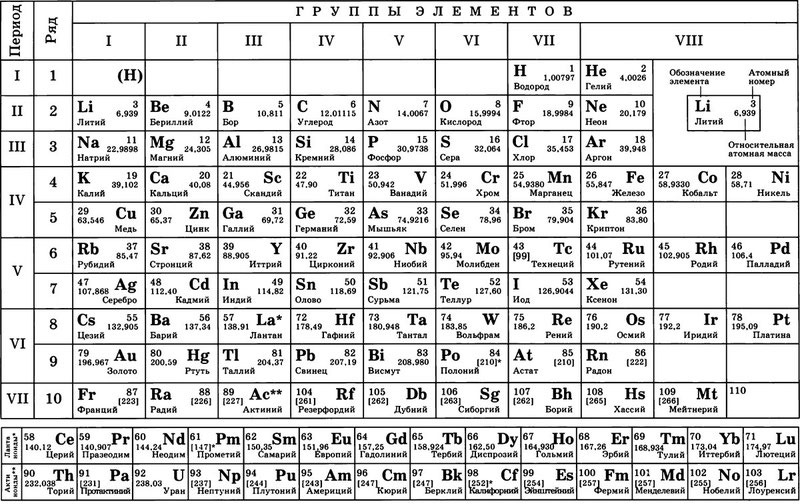
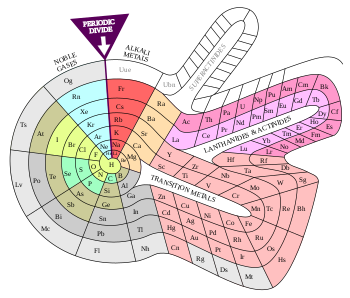
ТАБЛИЦА МЕНДЕЛЕЕВА — периодическая система химических элементов
Таблица Менделеева (периодическая система химических элементов) — это такая таблица, в которой классифицируются химические элементы по различным свойствам в зависимости от заряда их атомного ядра. Таблица является графическим изображением периодического закона, который открыл Дмитрий Иванович Менделеев в 1869 году. Изначальный вариант этой таблицы 1869 — 1871 гг. и устанавливал зависимость свойств элементов от их атомной массы. На данный момент элементы сводятся в двумерную таблицу, в которой каждый столбец — это группа, определяющая основные физико-химические свойства, а строки — это периоды, схожие друг с другом. Наиболее распространены 2 формы таблицы: короткая и длинная.
ТАБЛИЦА МЕНДЕЛЕЕВА

Порядковый номер элемента — это число равное числу протонов в ядре атома и числу электронов, которые вращаются вокруг него.
Чтобы посмотреть все свойства конкретного химического элемента нужно перейти по ссылке нажав на символ элемента в таблице.
| П. |
Группы химических элементов |
|||||||||
|
VIII |
||||||||||
| 1 |
1,00794 Водород |
4,0026 Гелий |
||||||||
| 2 |
6,941 Литий |
9,0122 Берилий |
10,811 Бор |
12,01115 Углерод |
14,0067 Азот |
15,9994 Кислород |
18,9984 Фтор |
20,179 Неон |
||
| 3 |
22,9898 Натрий |
24,305 Магний |
26,9815 Алюминий |
28,086 Кремний |
30,9738 Фосфор |
32,064 Сера |
35,454 Хлор |
39,948 Аргон |
||
| 4 |
39,0983 Калий |
40,08 Кальций |
44,956 Скандий |
47,88 Титан |
50,942 Ванадий |
51,996 Хром |
54,938 Марганец |
55,847 Железо |
58,9332 Кобальт |
58,69 Никель |
|
63,546 Медь |
65,39 Цинк |
69,72 Галлий |
72,61 Германий |
74,9216 Мышьяк |
78,96 Селен |
79,904 Бром |
83,80 Криптон |
|||
| 5 |
85,47 Рубидий |
87,62 Стронций |
88,906 Иттрий |
91,224 Цирконий |
92,906 Ниобий |
95,94 Молибден |
98,906 Технеций |
101,07 Рутений |
102,905 Родий |
106,42 Палладий |
|
107,868 Серебро |
112,41 Кадмий |
114,82 Индий |
118,71 Олово |
121,75 Сурьма |
127,60 Теллур |
126,9045 Йод |
131,30 Ксенон |
|||
| 6 |
132,905 Цезий |
137,327 Барий |
138,91 Лантан |
178,49 Гафний |
180,948 Тантал |
183,85 Вольфрам |
186,207 Рений |
190,2 Осмий |
192,22 Иридий |
195,09 Платина |
|
196,967 Золото |
200,59 Ртуть |
204,383 Таллий |
207,19 Свинец |
208,98 Висмут |
[209] Полоний |
[210] Астат |
[222] Радон |
|||
| 7 |
[223] Франций |
226,025 Радий |
[227] Актиний |
[261] Резерфордий |
[262] Дубний |
[263] Сиборгий |
[264] Борий |
[265] Хассий |
[266] Мейтнерий |
[281] Дармштадтий |
|
[281] Рентгений |
[285] Коперниций |
[284] Нихоний |
[289] Флеровий |
[288] Московий |
[293] Ливерморий |
[294] Теннесин |
[294] Оганесон |
|||
| 8 |
[316] Унуненний |
[320] Унбинилий |
||||||||
|
Высшие оксиды |
||||||||||
|
R2O |
RO |
R2O3 |
RO2 |
R2O5 |
RO3 |
R2O7 |
RO4 |
|||
|
Летучие водородные соединения |
||||||||||
|
RH4 |
RH3 |
H2R |
RH |
|
* ЛАНТАНОИДЫ |
|||||||||
|
114,16 Церий |
140,907 Празеодим |
144,24 Неодим |
[145] Прометий |
150,36 Самарий |
151,96 Европий |
157,25 Гадолиний |
158,924 Тербий |
162,5 Диспрозий |
164,93 Гольмий |
|
167,26 Эрбий |
168,94 Тулий |
173,04 Иттербий |
174,97 Лютеций |
||||||
|
** АКТИНОИДЫ |
|||||||||
|
232,038 Торий |
231,04 Протактиний |
238,03 Уран |
237,05 Нептуний |
[244] Плутоний |
[243] Америций |
[247] Кюрий |
[247] Берклий |
[251] Калифорний |
[254] Эйнштейний |
|
[257] Фермий |
[258] Менделевий |
[259] Нобелий |
260 Лоуренсий |
||||||
|
*** СУПЕРАКТИНОИДЫ |
|||||||||
|
320 Унбиуний |
* Унбибий |
* Унбитрий |
332 Унбиквадий |
* Унбипентий |
* Унбигексий |
||||
|
s – элементы |
p – элементы |
d – элементы |
f – элементы |
Периодическая система химических элементов Д.И. Менделеева
Расшифровка периодической системы химических элементов Д.И. Менделеева:
Периоды – горизонтальные строки химических элементов.
Группы – вертикальные столбцы химических элементов.
Подгруппы – А — главные (s- и р-элементы) и В — побочные (d- и f-элементы).
Номер периода – номер внешнего энергетического уровня в электронной формуле атома элемента.
Номер группы (для большинства элементов) – общее число валентных электронов (электронов внешнего энергетического уровня, а также предпоследнего d-подуровня, если он застроен не полностью).
Число элементов в периоде – максимальная емкость соответствующего энергетического уровня:
|
1 период |
2 элемента (1s2) |
5 период |
18 элементов (5s24d105p6) |
|
2 период |
8 элементов (2s22p6) |
6 период |
32 элемента (6s24f145d106p6) |
|
3 период |
8 элементов (3s23p6) |
7 период |
32 элемента (6s24f145d106p6) |
|
4 период |
18 элементов (4s23d104p6) |
8 период |
не завершен |
Построение периодов – в начале: два s-элемента, в конце: шесть р- элементов. В четвертом и пятом периодах между ними помещается по десять d-элементов, а в шестом и седьмом к ним добавляются четырнадцать f-элементов (формы электронных орбиталей).
В периоде – свойства химических элементов различаются между собой, т.к. электронные конфигурации валентных электронов их атомов различны.
В подгруппе – свойства элементов сходны между собой, т.к. электронные конфигурации валентных электронов их атомов сходны.
Причина периодичности свойств химических элементов заключается в периодической повторяемости сходных электронных конфигураций внешних энергетических уровней.
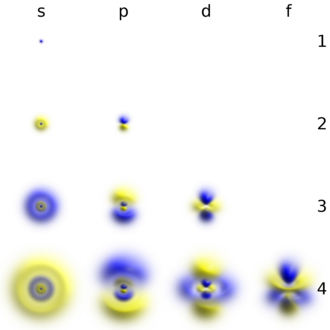
Формы электронных орбиталей (электронные семейства)
Классификация химических элементов по электронным конфигурациям их атомов (электронные орбитали)
|
Название семейства |
Тип конфигурации |
Застраиваемые подуровни |
|
s — элементы |
ns1–2 |
внешний (n) s-подуровень |
|
p -элементы |
ns2 np1–6 |
внешний (n) р-подуровень |
|
d — элементы |
(n-1)d1–10 ns1–2 |
предвнешний (n–1 ) d-подуровень |
|
f — элементы |
(n-2)f1–14 (n-1)d1–10 ns1–2 |
третий снаружи (n–2) f-подуровень |
Графическое изображение орбиталей
Свойства элементов таблицы Менделеева
Металлы – элементы главных подгрупп с числом валентных электронов от 1 до 3 (подгруппы IA, IIA, IIIА, кроме элемента бора), а также германий, олово, свинец, сурьма, висмут и полоний.
Неметаллы – бор и элементы главных подгрупп с числом валентных электронов от 4 до 7 (подгруппы IVA, VA, VIA, VIIA) кроме германия, олова, свинца, сурьмы, висмута и полония.
Переходные элементы – элементы побочных подгрупп (IB-VIIB); в виде простых веществ ведут себя как металлы.
Благородные газы – элементы подгруппы VIIIA, полностью застроенные энергетические подуровни s2p6, для гелия s2.
Галогены – элементы подгруппы VII(a) таблицы Менделеева, реагируют со всеми простыми веществами, кроме некот. неметаллов, являются энергичными окислителями, к ним относят F, Cl, Br, I, At, Ts.
Лантанойды – 15 элементов III группы 6-го периода, металлы с атомными номерами 57–71. Все они имеют стабильные изотопы, кроме прометия.
Актинойды – 15 радиоактивных элементов III группы 7-го периода с атомными номерами 89–103.
Свойства элементов в подгруппах закономерно изменяются сверху вниз:
— металлические свойства усиливаются и неметаллические свойства ослабевают;
— атомный радиус увеличивается;
— возрастает сила образованных элементом оснований и бескислородных кислот;
— уменьшается электроотрицательность.
В периодах с увеличением порядкового номера элемента прослеживается следующая закономерность:
— увеличивается электроотрицательность;
— металлические свойства ослабевают, неметаллические усиливаются;
— уменьшается атомный радиус.
Все элементы таблицы Менделеева, исключая гелий, неон и аргон, образуют кислородные соединения, которые изображены общими формулами под каждой группой в порядке возрастания степени окисления элементов: R2O, RO, R2O3, RO2, R2O5, RO3, R2O7, RO4, где R — обозначает элемент группы.
Элементы главных подгрупп, начиная с IV группы, образуют газообразные водородные соединения: RH4, RH3, RH2, RH. Соединения RH4 имеют нейтральный характер; RH3 – слабоосновной; RH2 – слабокислый; RH – сильнокислый характер.
История открытия периодического закона Менделеевым Д.И.
Самый важный вклад в систематизацию химических элементов внёс русский выдающийся химик Дмитрий Иванович Менделеев, автор труда «Основы химии», который в марте 1869 года представил Русскому химическому обществу (РХО) периодический закон химических элементов, изложенный в нескольких основных положениях.
В 1871 году Менделеев в итоговой статье «Периодическая законность химических элементов» дал формулировку Периодического закона: «Свойства элементов, а потому и свойства образуемых ими простых и сложных тел стоят в периодической зависимости от атомного веса». Тогда же Менделеев придал своей периодической таблице классический вид (короткая таблица, смотрите ниже).
В современном изложении периодический закон химических элементов звучит так: «Свойства простых веществ, а также свойства и формы соединений элементов находятся в периодической зависимости от заряда ядра атомов элементов (порядкового номера).»
Периодическая таблица элементов Менделеева длинная форма
Длинная форма таблицы Менделеева (или длиннопериодная форма) состоит из 18 групп с лева на право от щелочных металов до благородных газов. считается официальной версией с 1989 года.
Таблица Менделеева для печати в хорошем качестве скачать
Вы можете скачать таблицу Менделеева на выбор короткую или длинную форму в цветном и черно-белом цвете, для этого откройте по ссылке ниже изображение и сохраните его себе на компьютер.
1) Таблица Менделеева для печати в хорошем качестве цветная (короткая форма)
2) Таблица Менделеева для печати в хорошем качестве черно-белая (короткая форма)
3) Таблица Менделеева для печати в хорошем качестве цветная (длинная форма)
4) Таблица Менделеева для печати в хорошем качестве черно-белая (длинная форма)
____________
Источник информации:
1. Большой химический справочник / А.И.Волков, — М.: 2005.
2. Большая энциклопедия химических элементов. Периодическая таблица Менделеева / И.А.Леенсон. — Москва : 2014.
3. По материалам сайта ru.wikipedia.org
Periodic table of the chemical elements
The periodic table, also known as the periodic table of the (chemical) elements, is a rows and columns arrangement of the chemical elements. It is widely used in chemistry, physics, and other sciences, and is generally seen as an icon of chemistry. It is a graphic formulation of the periodic law, which states that the properties of the chemical elements exhibit an approximate periodic dependence on their atomic numbers.
The table is divided into four roughly rectangular areas called blocks. The rows of the table are called periods, and the columns are called groups. Elements from the same group of the periodic table show similar chemical characteristics. Trends run through the periodic table, with nonmetallic character (keeping their own electrons) increasing from left to right across a period, and from down to up across a group, and metallic character (surrendering electrons to other atoms) increasing in the opposite direction. The underlying reason for these trends is electron configurations of atoms. The periodic table exclusively lists electrically neutral atoms that have an equal number of positively charged protons and negatively charged electrons and puts isotopes (atoms with the same number of protons but different numbers of neutrons) at the same place. Other atoms, like nuclides and isotopes, are graphically collected in other tables like the tables of nuclides (often called Segrè charts).
The first periodic table to become generally accepted was that of the Russian chemist Dmitri Mendeleev in 1869: he formulated the periodic law as a dependence of chemical properties on atomic mass. Because not all elements were then known, there were gaps in his periodic table, and Mendeleev successfully used the periodic law to predict properties of some of the missing elements. The periodic law was recognized as a fundamental discovery in the late 19th century, and it was explained with the discovery of the atomic number and pioneering work in quantum mechanics of the early 20th century that illuminated the internal structure of the atom. With Glenn T. Seaborg’s 1945 discovery that the actinides were in fact f-block rather than d-block elements, a recognisably modern form of the table was reached. The periodic table and law are now a central and indispensable part of modern chemistry.
The periodic table continues to evolve with the progress of science. In nature, only elements up to atomic number 94 exist; to go further, it was necessary to synthesise new elements in the laboratory. Today, all the first 118 elements are known, completing the first seven rows of the table, but chemical characterisation is still needed for the heaviest elements to confirm that their properties match their positions. It is not yet known how far the table will stretch beyond these seven rows and whether the patterns of the known part of the table will continue into this unknown region. Some scientific discussion also continues regarding whether some elements are correctly positioned in today’s table. Many alternative representations of the periodic law exist, and there is some discussion as to whether there is an optimal form of the periodic table.
Overview
The periodic table is a 2-dimensional structured table. The elements are placed in table cells, in reading order of ascending atomic number. The table is divided into four blocks, reflecting the filling of electrons into types of subshell. The table columns are called groups, and the rows are called periods. New periods begin when a new electron shell starts to fill: elements in the same group have the same number of electrons that can be used for chemistry (except for helium in the noble gas group), so that similar physical and chemical properties recur at regular intervals.
- v
- t
- e
Atomic structure
Main article: Atom
3D views of some hydrogen-like atomic orbitals showing probability density and phase (g orbitals and higher are not shown)
The smallest constituents of all normal matter are known as atoms. Atoms are extremely small, being about one ten-billionth of a meter across; thus their internal structure is governed by quantum mechanics.[3] Atoms consist of a small positively charged nucleus, made of positively charged protons and uncharged neutrons, surrounded by a cloud of negatively charged electrons; the charges cancel out, so atoms are neutral.[4] Electrons participate in chemical reactions, but the nucleus does not.[4] When atoms participate in chemical reactions, they either gain or lose electrons to form positively- or negatively-charged ions; or share electrons with each other.[5]
Atoms can be subdivided into different types based on the number of protons (and thus also electrons) they have.[4] This is called the atomic number, often symbolised Z[6] (for «Zahl» — German for «number»). Each distinct atomic number therefore corresponds to a class of atom: these classes are called the chemical elements.[7] The chemical elements are what the periodic table classifies and organises. Hydrogen is the element with atomic number 1; helium, atomic number 2; lithium, atomic number 3; and so on. Each of these names can be further abbreviated by a one- or two-letter chemical symbol; those for hydrogen, helium, and lithium are respectively H, He, and Li.[8] Neutrons do not affect the atom’s chemical identity, but do affect its weight. Atoms with the same number of protons but different numbers of neutrons are called isotopes of the same chemical element.[8] Naturally occurring elements usually occur as mixes of different isotopes; since each isotope usually occurs with a characteristic abundance, naturally occurring elements have well-defined atomic weights, defined as the average mass of a naturally occurring atom of that element.[9]
Today, 118 elements are known, the first 94 of which are known to occur naturally on Earth at present.[10][a] Of the 94 natural elements, eighty have a stable isotope and one more (bismuth) has an almost-stable isotope (with a half-life over a billion times the age of the universe).[14] Two more, thorium and uranium, have isotopes undergoing radioactive decay with a half-life comparable to the age of the Earth. The stable elements plus bismuth, thorium, and uranium make up the 83 primordial elements that survived from the Earth’s formation.[b] The remaining eleven natural elements decay quickly enough that their continued trace occurrence rests primarily on being constantly regenerated as intermediate products of the decay of thorium and uranium.[c] All 24 known artificial elements are radioactive.[8]
Electron configuration
The periodic table is a graphic description of the periodic law,[18] which states that the properties and atomic structures of the chemical elements are a periodic function of their atomic number.[19] Elements are placed in the periodic table by their electron configurations,[20] which exhibit periodic recurrences that explain the trends of properties across the periodic table.[21]
An electron can be thought of as inhabiting an atomic orbital, which characterises the probability it can be found in any particular region of the atom. Their energies are quantised, which is to say that they can only take discrete values. Furthermore, electrons obey the Pauli exclusion principle: different electrons must always be in different states. This allows classification of the possible states an electron can take in various energy levels known as shells, divided into individual subshells, which each contain one or more orbitals. Each orbital can contain up to two electrons: they are distinguished by a quantity known as spin, conventionally labeled «up» or «down».[22][d] In a cold atom (one in its ground state), electrons arrange themselves in such a way that the total energy they have is minimised by occupying the lowest-energy orbitals available.[24] Only the outermost electrons (so-called valence electrons) have enough energy to break free of the nucleus and participate in chemical reactions with other atoms. The others are called core electrons.[25]
| ℓ → n ↓ |
0 | 1 | 2 | 3 | 4 | 5 | 6 | |
|---|---|---|---|---|---|---|---|---|
| Orbital | s | p | d | f | g | h | i | Capacity of shell (2n2)[26] |
| 1 | 1s | 2 | ||||||
| 2 | 2s | 2p | 8 | |||||
| 3 | 3s | 3p | 3d | 18 | ||||
| 4 | 4s | 4p | 4d | 4f | 32 | |||
| 5 | 5s | 5p | 5d | 5f | 5g | 50 | ||
| 6 | 6s | 6p | 6d | 6f | 6g | 6h | 72 | |
| 7 | 7s | 7p | 7d | 7f | 7g | 7h | 7i | 98 |
| Capacity of subshell | 2 | 6 | 10 | 14 | 18 | 22 | 26 |
Elements are known with up to the first seven shells occupied. The first shell contains only one orbital, a spherical s orbital. As it is in the first shell, this is called the 1s orbital. This can hold up to two electrons. The second shell similarly contains a 2s orbital, and it also contains three dumbbell-shaped 2p orbitals, and can thus fill up to eight electrons (2×1 + 2×3 = 8). The third shell contains one 3s orbital, three 3p orbitals, and five 3d orbitals, and thus has a capacity of 2×1 + 2×3 + 2×5 = 18. The fourth shell contains one 4s orbital, three 4p orbitals, five 4d orbitals, and seven 4f orbitals, thus leading to a capacity of 2×1 + 2×3 + 2×5 + 2×7 = 32.[27] Higher shells contain more types of orbitals that continue the pattern, but such types of orbitals are not filled in the ground states of known elements.[28] The subshell types are characterised by the quantum numbers. Four numbers describe an orbital in an atom completely: the principal quantum number n, the azimuthal quantum number ℓ (the orbital type), the magnetic quantum number mℓ, and the spin quantum number s.[21]
The order of subshell filling
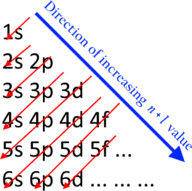
Idealized order of subshell filling according to the Madelung rule
The sequence in which the subshells are filled is given in most cases by the Aufbau principle, also known as the Madelung or Klechkovsky rule (after Erwin Madelung and Vsevolod Klechkovsky respectively). This rule was first observed empirically by Madelung, and Klechkovsky and later authors gave it theoretical justification.[29][30][31][32] The shells overlap in energies, and the Madelung rule specifies the sequence of filling according to:[30]
- 1s ≪ 2s < 2p ≪ 3s < 3p ≪ 4s < 3d < 4p ≪ 5s < 4d < 5p ≪ 6s < 4f < 5d < 6p ≪ 7s < 5f < 6d < 7p ≪ …
Here the sign ≪ means «much less than» as opposed to < meaning just «less than».[30] Phrased differently, electrons enter orbitals in order of increasing n + ℓ, and if two orbitals are available with the same value of n + ℓ, the one with lower n is occupied first.[28][32] In general, orbitals with the same value of n + ℓ are similar in energy, but in the case of the s-orbitals (with ℓ = 0), quantum effects raise their energy to approach that of the next n + ℓ group. Hence the periodic table is usually drawn to begin each row (often called a period) with the filling of a new s-orbital, which corresponds to the beginning of a new shell.[30][31][27] Thus, with the exception of the first row, each period length appears twice:[30]
- 2, 8, 8, 18, 18, 32, 32, …
The overlaps get quite close at the point where the d-orbitals enter the picture,[33] and the order can shift slightly with atomic number[34] and atomic charge.[35][e]
Starting from the simplest atom, this lets us build up the periodic table one at a time in order of atomic number, by considering the cases of single atoms. In hydrogen, there is only one electron, which must go in the lowest-energy orbital 1s. This electron configuration is written 1s1, where the superscript indicates the number of electrons in the subshell. Helium adds a second electron, which also goes into 1s, completely filling the first shell and giving the configuration 1s2.[21][38][f]
Starting from the third element, lithium, the first shell is full, so its third electron occupies a 2s orbital, giving a 1s2 2s1 configuration. The 2s electron is lithium’s only valence electron, as the 1s subshell is now too tightly bound to the nucleus to participate in chemical bonding to other atoms. Thus the filled first shell is called a «core shell» for this and all heavier elements. The 2s subshell is completed by the next element beryllium (1s2 2s2). The following elements then proceed to fill the 2p subshell. Boron (1s2 2s2 2p1) puts its new electron in a 2p orbital; carbon (1s2 2s2 2p2) fills a second 2p orbital; and with nitrogen (1s2 2s2 2p3) all three 2p orbitals become singly occupied. This is consistent with Hund’s rule, which states that atoms usually prefer to singly occupy each orbital of the same type before filling them with the second electron. Oxygen (1s2 2s2 2p4), fluorine (1s2 2s2 2p5), and neon (1s2 2s2 2p6) then complete the already singly filled 2p orbitals; the last of these fills the second shell completely.[21][38]
Starting from element 11, sodium, the second shell is full, making the second shell a core shell for this and all heavier elements. The eleventh electron begins the filling of the third shell by occupying a 3s orbital, giving a configuration of 1s2 2s2 2p6 3s1 for sodium. This configuration is abbreviated [Ne] 3s1, where [Ne] represents neon’s configuration. Magnesium ([Ne] 3s2) finishes this 3s orbital, and the following six elements aluminium, silicon, phosphorus, sulfur, chlorine, and argon fill the three 3p orbitals ([Ne] 3s2 3p1 through [Ne] 3s2 3p6).[21][38] This creates an analogous series in which the outer shell structures of sodium through argon are analogous to those of lithium through neon, and is the basis for the periodicity of chemical properties that the periodic table illustrates:[21] at regular but changing intervals of atomic numbers, the properties of the chemical elements approximately repeat.[18]
The first eighteen elements can thus be arranged as the start of a periodic table. Elements in the same column have the same number of valence electrons and have analogous valence electron configurations: these columns are called groups. The single exception is helium, which has two valence electrons like beryllium and magnesium, but is typically placed in the column of neon and argon to emphasise that its outer shell is full. (Some contemporary authors question even this single exception, preferring to consistently follow the valence configurations and place helium over beryllium.) There are eight columns in this periodic table fragment, corresponding to at most eight outer-shell electrons.[5] A period begins when a new shell starts filling.[27] Finally, the colouring illustrates the blocks: the elements in the s-block (coloured red) are filling s-orbitals, while those in the p-block (coloured yellow) are filling p-orbitals.[27]
| 1 H |
2 He |
2×1 = 2 elements 1s 0p |
||||||
| 3 Li |
4 Be |
5 B |
6 C |
7 N |
8 O |
9 F |
10 Ne |
2×(1+3) = 8 elements 2s 2p |
| 11 Na |
12 Mg |
13 Al |
14 Si |
15 P |
16 S |
17 Cl |
18 Ar |
2×(1+3) = 8 elements 3s 3p |
Starting the next row, for potassium and calcium the 4s subshell is the lowest in energy, and therefore they fill it.[21][38] Potassium adds one electron to the 4s shell ([Ar] 4s1), and calcium then completes it ([Ar] 4s2). However, starting from scandium ([Ar] 3d1 4s2) the 3d subshell becomes the next highest in energy. The 4s and 3d subshells have approximately the same energy and they compete for filling the electrons, and so the occupation is not quite consistently filling the 3d orbitals one at a time. The precise energy ordering of 3d and 4s changes along the row, and also changes depending on how many electrons are removed from the atom. For example, due to the repulsion between the 3d electrons and the 4s ones, at chromium the 4s energy level becomes slightly higher than 3d, and so it becomes more profitable to have a [Ar] 3d5 4s1 configuration than an [Ar] 3d4 4s2 one. A similar anomaly occurs at copper.[21] These are violations of the Madelung rule. Such anomalies however do not have any chemical significance,[35] as the various configurations are so close in energy to each other[33] that the presence of a nearby atom can shift the balance.[21] The periodic table therefore ignores these and considers only idealised configurations.[20]
At zinc ([Ar] 3d10 4s2), the 3d orbitals are completely filled with a total of ten electrons.[21][38] Next come the 4p orbitals, completing the row, which are filled progressively by gallium ([Ar] 3d10 4s2 4p1) through krypton ([Ar] 3d10 4s2 4p6), in a manner analogous to the previous p-block elements.[21][38] From gallium onwards, the 3d orbitals form part of the electronic core, and no longer participate in chemistry.[37] The s- and p-block elements, which fill their outer shells, are called main-group elements; the d-block elements (coloured blue below), which fill an inner shell, are called transition elements (or transition metals, since they are all metals).[41]
The next eighteen elements fill the 5s orbitals (rubidium and strontium), then 4d (yttrium through cadmium, again with a few anomalies along the way), and then 5p (indium through xenon).[27][38] Again, from indium onward the 4d orbitals are in the core.[38][42] Hence the fifth row has the same structure as the fourth.[27]
| 1 H |
2 He |
2×1 = 2 elements 1s 0d 0p |
||||||||||||||||
| 3 Li |
4 Be |
5 B |
6 C |
7 N |
8 O |
9 F |
10 Ne |
2×(1+3) = 8 elements 2s 0d 2p |
||||||||||
| 11 Na |
12 Mg |
13 Al |
14 Si |
15 P |
16 S |
17 Cl |
18 Ar |
2×(1+3) = 8 elements 3s 0d 3p |
||||||||||
| 19 K |
20 Ca |
21 Sc |
22 Ti |
23 V |
24 Cr |
25 Mn |
26 Fe |
27 Co |
28 Ni |
29 Cu |
30 Zn |
31 Ga |
32 Ge |
33 As |
34 Se |
35 Br |
36 Kr |
2×(1+3+5) = 18 elements 4s 3d 4p |
| 37 Rb |
38 Sr |
39 Y |
40 Zr |
41 Nb |
42 Mo |
43 Tc |
44 Ru |
45 Rh |
46 Pd |
47 Ag |
48 Cd |
49 In |
50 Sn |
51 Sb |
52 Te |
53 I |
54 Xe |
2×(1+3+5) = 18 elements 5s 4d 5p |
The sixth row of the table likewise starts with two s-block elements: caesium and barium.[38] After this, the first f-block elements (coloured green below) begin to appear, starting with lanthanum. These are sometimes termed inner transition elements.[41] As there are now not only 4f but also 5d and 6s subshells at similar energies, competition occurs once again with many irregular configurations;[33] this resulted in some dispute about where exactly the f-block is supposed to begin, but most who study the matter agree that it starts at lanthanum in accordance with the Aufbau principle.[43] Even though lanthanum does not itself fill the 4f subshell as a single atom, because of repulsion between electrons,[35] its 4f orbitals are low enough in energy to participate in chemistry.[44] At ytterbium, the seven 4f orbitals are completely filled with fourteen electrons; thereafter, a series of ten transition elements (lutetium through mercury) follows,[38][45][46][47] and finally six main-group elements (thallium through radon) complete the period.[38][48] From lutetium onwards the 4f orbitals are in the core,[38] and from thallium onwards so are the 5d orbitals.[38][37][49]
The seventh row is analogous to the sixth row: 7s fills (francium and radium), then 5f (actinium to nobelium), then 6d (lawrencium to copernicium), and finally 7p (nihonium to oganesson).[38] Starting from lawrencium the 5f orbitals are in the core,[38] and probably the 6d orbitals join the core starting from nihonium.[38][50][g] Again there are a few anomalies along the way:[27] for example, as single atoms neither actinium nor thorium actually fills the 5f subshell, and lawrencium does not fill the 6d shell, but all these subshells can still become filled in chemical environments.[52][53][54] For a very long time, the seventh row was incomplete as most of its elements do not occur in nature. The missing elements beyond uranium started to be synthesised in the laboratory in 1940, when neptunium was made.[55] The row was completed with the synthesis of tennessine in 2010[56] (the last element oganesson had already been made in 2002),[57] and the last elements in this seventh row were given names in 2016.[58]
| 1 H |
2 He |
2×1 = 2 elements 1s 0f 0d 0p |
||||||||||||||||||||||||||||||
| 3 Li |
4 Be |
5 B |
6 C |
7 N |
8 O |
9 F |
10 Ne |
2×(1+3) = 8 elements 2s 0f 0d 2p |
||||||||||||||||||||||||
| 11 Na |
12 Mg |
13 Al |
14 Si |
15 P |
16 S |
17 Cl |
18 Ar |
2×(1+3) = 8 elements 3s 0f 0d 3p |
||||||||||||||||||||||||
| 19 K |
20 Ca |
21 Sc |
22 Ti |
23 V |
24 Cr |
25 Mn |
26 Fe |
27 Co |
28 Ni |
29 Cu |
30 Zn |
31 Ga |
32 Ge |
33 As |
34 Se |
35 Br |
36 Kr |
2×(1+3+5) = 18 elements 4s 0f 3d 4p |
||||||||||||||
| 37 Rb |
38 Sr |
39 Y |
40 Zr |
41 Nb |
42 Mo |
43 Tc |
44 Ru |
45 Rh |
46 Pd |
47 Ag |
48 Cd |
49 In |
50 Sn |
51 Sb |
52 Te |
53 I |
54 Xe |
2×(1+3+5) = 18 elements 5s 0f 4d 5p |
||||||||||||||
| 55 Cs |
56 Ba |
57 La |
58 Ce |
59 Pr |
60 Nd |
61 Pm |
62 Sm |
63 Eu |
64 Gd |
65 Tb |
66 Dy |
67 Ho |
68 Er |
69 Tm |
70 Yb |
71 Lu |
72 Hf |
73 Ta |
74 W |
75 Re |
76 Os |
77 Ir |
78 Pt |
79 Au |
80 Hg |
81 Tl |
82 Pb |
83 Bi |
84 Po |
85 At |
86 Rn |
2×(1+3+5+7) = 32 elements 6s 4f 5d 6p |
| 87 Fr |
88 Ra |
89 Ac |
90 Th |
91 Pa |
92 U |
93 Np |
94 Pu |
95 Am |
96 Cm |
97 Bk |
98 Cf |
99 Es |
100 Fm |
101 Md |
102 No |
103 Lr |
104 Rf |
105 Db |
106 Sg |
107 Bh |
108 Hs |
109 Mt |
110 Ds |
111 Rg |
112 Cn |
113 Nh |
114 Fl |
115 Mc |
116 Lv |
117 Ts |
118 Og |
2×(1+3+5+7) = 32 elements 7s 5f 6d 7p |
This completes the modern periodic table, with all seven rows completely filled to capacity.[58]
Electron configuration table
The following table shows the electron configuration of a neutral gas-phase atom of each element. Different configurations can be favoured in different chemical environments.[35] The main-group elements have entirely regular electron configurations; the transition and inner transition elements show twenty irregularities due to the aforementioned competition between subshells close in energy level. For the last ten elements (109–118), experimental data is lacking[59] and therefore calculated configurations have been shown instead.[60] Completely filled subshells have been greyed out.
Electron configurations of the chemical elements (neutral gaseous atoms in the ground state) |
||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Group: | 1 | 2 | | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | |||||||||||||
| 1s: | 1 H 1 |
2 He 2 |
||||||||||||||||||||||||||||||
| [He] 2s: 2p: |
3 Li 1 — |
4 Be 2 — |
5 B 2 1 |
6 C 2 2 |
7 N 2 3 |
8 O 2 4 |
9 F 2 5 |
10 Ne 2 6 |
||||||||||||||||||||||||
| [Ne] 3s: 3p: |
11 Na 1 — |
12 Mg 2 — |
13 Al 2 1 |
14 Si 2 2 |
15 P 2 3 |
16 S 2 4 |
17 Cl 2 5 |
18 Ar 2 6 |
||||||||||||||||||||||||
| [Ar] 4s: 3d: 4p: |
19 K 1 — — |
20 Ca 2 — — |
21 Sc 2 1 — |
22 Ti 2 2 — |
23 V 2 3 — |
24 Cr 1 5 — |
25 Mn 2 5 — |
26 Fe 2 6 — |
27 Co 2 7 — |
28 Ni 2 8 — |
29 Cu 1 10 — |
30 Zn 2 10 — |
31 Ga 2 10 1 |
32 Ge 2 10 2 |
33 As 2 10 3 |
34 Se 2 10 4 |
35 Br 2 10 5 |
36 Kr 2 10 6 |
||||||||||||||
| [Kr] 5s: 4d: 5p: |
37 Rb 1 — — |
38 Sr 2 — — |
39 Y 2 1 — |
40 Zr 2 2 — |
41 Nb 1 4 — |
42 Mo 1 5 — |
43 Tc 2 5 — |
44 Ru 1 7 — |
45 Rh 1 8 — |
46 Pd — 10 — |
47 Ag 1 10 — |
48 Cd 2 10 — |
49 In 2 10 1 |
50 Sn 2 10 2 |
51 Sb 2 10 3 |
52 Te 2 10 4 |
53 I 2 10 5 |
54 Xe 2 10 6 |
||||||||||||||
| [Xe] 6s: 4f: 5d: 6p: |
55 Cs 1 — — — |
56 Ba 2 — — — |
57 La 2 — 1 — |
58 Ce 2 1 1 — |
59 Pr 2 3 — — |
60 Nd 2 4 — — |
61 Pm 2 5 — — |
62 Sm 2 6 — — |
63 Eu 2 7 — — |
64 Gd 2 7 1 — |
65 Tb 2 9 — — |
66 Dy 2 10 — — |
67 Ho 2 11 — — |
68 Er 2 12 — — |
69 Tm 2 13 — — |
70 Yb 2 14 — — |
71 Lu 2 14 1 — |
72 Hf 2 14 2 — |
73 Ta 2 14 3 — |
74 W 2 14 4 — |
75 Re 2 14 5 — |
76 Os 2 14 6 — |
77 Ir 2 14 7 — |
78 Pt 1 14 9 — |
79 Au 1 14 10 — |
80 Hg 2 14 10 — |
81 Tl 2 14 10 1 |
82 Pb 2 14 10 2 |
83 Bi 2 14 10 3 |
84 Po 2 14 10 4 |
85 At 2 14 10 5 |
86 Rn 2 14 10 6 |
| [Rn] 7s: 5f: 6d: 7p: |
87 Fr 1 — — — |
88 Ra 2 — — — |
89 Ac 2 — 1 — |
90 Th 2 — 2 — |
91 Pa 2 2 1 — |
92 U 2 3 1 — |
93 Np 2 4 1 — |
94 Pu 2 6 — — |
95 Am 2 7 — — |
96 Cm 2 7 1 — |
97 Bk 2 9 — — |
98 Cf 2 10 — — |
99 Es 2 11 — — |
100 Fm 2 12 — — |
101 Md 2 13 — — |
102 No 2 14 — — |
103 Lr 2 14 — 1 |
104 Rf 2 14 2 — |
105 Db 2 14 3 — |
106 Sg 2 14 4 — |
107 Bh 2 14 5 — |
108 Hs 2 14 6 — |
109 Mt 2 14 7 — |
110 Ds 2 14 8 — |
111 Rg 2 14 9 — |
112 Cn 2 14 10 — |
113 Nh 2 14 10 1 |
114 Fl 2 14 10 2 |
115 Mc 2 14 10 3 |
116 Lv 2 14 10 4 |
117 Ts 2 14 10 5 |
118 Og 2 14 10 6 |
|
Group names and numbers
Under an international naming convention, the groups are numbered numerically from 1 to 18 from the leftmost column (the alkali metals) to the rightmost column (the noble gases). The f-block groups are ignored in this numbering.[61] Groups can also be named by their first element, e.g. the «scandium group» for group 3.[61] Previously, groups were known by Roman numerals. In America, the Roman numerals were followed by either an «A» if the group was in the s- or p-block, or a «B» if the group was in the d-block. The Roman numerals used correspond to the last digit of today’s naming convention (e.g. the group 4 elements were group IVB, and the group 14 elements were group IVA). In Europe, the lettering was similar, except that «A» was used if the group was before group 10, and «B» was used for groups including and after group 10. In addition, groups 8, 9 and 10 used to be treated as one triple-sized group, known collectively in both notations as group VIII. In 1988, the new IUPAC (International Union of Pure and Applied Chemistry) naming system (1–18) was put into use, and the old group names (I–VIII) were deprecated.[62]
- v
- t
- e
| IUPAC group | 1a | 2 | —b | 3c | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mendeleev (I–VIII) | IA | IIA | IIIB | IVB | VB | VIB | VIIB | VIIIB | IB | IIB | IIIB | IVB | VB | VIB | VIIB | d | |||
| CAS (US, A-B-A) | IA | IIA | IIIB | IVB | VB | VIB | VIIB | VIIIB | IB | IIB | IIIA | IVA | VA | VIA | VIIA | VIIIA | |||
| Old IUPAC (Europe, A-B) | IA | IIA | IIIA | IVA | VA | VIA | VIIA | VIIIB | IB | IIB | IIIB | IVB | VB | VIB | VIIB | 0 | |||
| Trivial namer | H and alkali metals | alkaline earth metals | triels | tetrels | pnictogens | chalcogens | halogens | noble gases | |||||||||||
| Name by elementr | lithium group | beryllium group | scandium group | titanium group | vanadium group | chromium group | manganese group | iron group | cobalt group | nickel group | copper group | zinc group | boron group | carbon group | nitrogen group | oxygen group | fluorine group | helium or neon group | |
| Period 1 | H | He | |||||||||||||||||
| Period 2 | Li | Be | B | C | N | O | F | Ne | |||||||||||
| Period 3 | Na | Mg | Al | Si | P | S | Cl | Ar | |||||||||||
| Period 4 | K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | |
| Period 5 | Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | |
| Period 6 | Cs | Ba | La–Yb | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| Period 7 | Fr | Ra | Ac–No | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
a Group 1 is composed of hydrogen (H) and the alkali metals. Elements of the group have one s-electron in the outer electron shell. Hydrogen is not considered to be an alkali metal as it is not a metal, though it is more analogous to them than any other group. This makes the group somewhat exceptional.
b The 14 f-block groups (columns) do not have a group number.
c The correct composition of group 3 is scandium (Sc), yttrium (Y), lutetium (Lu), and lawrencium (Lr), as shown here: this is endorsed by 1988[62] and 2021[63] IUPAC reports on the question. General inorganic chemistry texts often put scandium (Sc), yttrium (Y), lanthanum (La), and actinium (Ac) in group 3, so that Ce–Lu and Th–Lr become the f-block between groups 3 and 4; this was based on incorrectly measured electron configurations from history,[64] and Lev Landau and Evgeny Lifshitz already considered it incorrect in 1948.[65] Arguments can still occasionally be encountered in the contemporary literature purporting to defend it, but most authors consider them logically inconsistent.[43][39][66] Some sources follow a compromise that puts La–Lu and Ac–Lr as the f-block rows (despite that giving 15 f-block elements in each row, which contradicts quantum mechanics), leaving the heavier members of group 3 ambiguous.[63] See also Group 3 element#Composition.
d Group 18, the noble gases, were not discovered at the time of Mendeleev’s original table. Later (1902), Mendeleev accepted the evidence for their existence, and they could be placed in a new «group 0», consistently and without breaking the periodic table principle.
r Group name as recommended by IUPAC.
Presentation forms
For reasons of space,[27][67] the periodic table is commonly presented with the f-block elements cut out and positioned placed as a distinct part below the main body.[5][27][62] It reduces the number of element columns from 32 to 18.[27]
Both forms represent the same periodic table.[8] The form with the f-block included in the main body is sometimes called the 32-column[8] or long form;[68] the form with the f-block cut out the 18-column[8] or medium-long form.[68] The 32-column form has the advantage of showing all elements in their correct sequence, but it has the disadvantage of requiring more space.[69] The form chosen is an editorial choice, and does not imply any change of scientific claim or statement. For example, when discussing the composition of group 3, the options can be shown equally (unprejudiced) in both forms.[70]
Periodic tables usually at least show the elements’ symbols; many also provide supplementary information about the elements, either via colour-coding or as data in the cells. The above table shows the names and atomic numbers of the elements, and also their blocks, natural occurrences and standard atomic weights. For the short-lived elements without standard atomic weights, the mass number of the most stable known isotope is used instead. Other tables may include properties such as state of matter, melting and boiling points, densities, as well as provide different classifications of the elements.[h]
Variations
Period 1
Although the modern periodic table is standard today, the placement of the period 1 elements hydrogen and helium remains an open issue under discussion, and some variation can be found.[37][71] Following electron configurations, hydrogen would be placed in group 1, and helium would be placed in group 2.[37] The group 1 placement of hydrogen is common, but helium is almost always placed in group 18 with the other noble gases.[8] The debate has to do with conflicting understandings of whether chemical or electronic properties should primarily decide periodic table placement, and conflicting views of how the evidence should be used.[71]
Like the group 1 metals, hydrogen has one electron in its outermost shell[72] and typically loses its only electron in chemical reactions.[73] It has some metal-like chemical properties, being able to displace some metals from their salts.[73] But hydrogen forms a diatomic nonmetallic gas at standard conditions, unlike the alkali metals which are reactive solid metals. This and hydrogen’s formation of hydrides, in which it gains an electron, brings it close to the properties of the halogens which do the same[73] (though it is rarer for hydrogen to form H− than H+).[74] Moreover, the lightest two halogens (fluorine and chlorine) are gaseous like hydrogen at standard conditions.[73] Some properties of hydrogen are not a good fit for either group: hydrogen is neither highly oxidising nor highly reducing and is not reactive with water.[74] Hydrogen thus has properties corresponding to both those of the alkali metals and the halogens, but matches neither group perfectly, and is thus difficult to place by its chemistry.[73] Therefore, while the electronic placement of hydrogen in group 1 predominates, some rarer arrangements show either hydrogen in group 17,[75] duplicate hydrogen in both groups 1 and 17,[76][77] or float it separately from all groups.[77][78][37] This last option has nonetheless been criticised by the chemist and philosopher of science Eric Scerri on the grounds that it appears to imply that hydrogen is above the periodic law altogether, unlike all the other elements.[79]
Helium is an unreactive noble gas at standard conditions, and has a full outer shell: these properties are like the noble gases in group 18, but not at all like the reactive alkaline earth metals of group 2. Therefore, helium is nearly universally placed in group 18[8] which its properties best match:[37] a proposal to move helium to group 2 was rejected by IUPAC in 1988 for these reasons.[62] However, helium only has two electrons in its outermost shell, whereas the other noble gases have eight; and it is an s-block element, whereas all other noble gases are p-block elements. Also, solid helium crystallises in a hexagonal close-packed structure, which matches beryllium and magnesium in group 2, but not the other noble gases in group 18.[80] In these ways helium better matches the alkaline earth metals[72][37] and it is occasionally placed to head group 2 today.[81] With recent theoretical developments in light noble gas chemistry, in which helium is expected to show less inertness than neon and to form (HeO)(LiF)2 with a structure similar to the analogous beryllium compound (but with no expected neon analogue), more chemists have begun to advocate placing helium in group 2.[82][83][84][85] The first-row anomaly trend is often cited in support of this reassignment, as helium as the first s2 element before the alkaline earth metals stands out as anomalous in a way that helium as the first noble gas does not.[82] Thus for example a large difference in atomic radii between the first and second members of each main group is seen in groups 1 and 13–17: it exists between neon and argon, and between helium and beryllium, but not between helium and neon. Moving helium to group 2 makes this trend consistent in groups 2 and 18 as well.[86][87] Tables that float both hydrogen and helium outside all groups may also rarely be encountered.[78][37][38]
Group 3
In many periodic tables, the f-block is shifted one element to the right, so that lanthanum and actinium become d-block elements in group 3, and Ce–Lu and Th–Lr form the f-block tearing the d-block into two very uneven portions. This is a holdover from early mistaken measurements of electron configurations.[64] The 4f shell is completely filled at ytterbium, and for that reason Lev Landau and Evgeny Lifshitz in 1948 considered it incorrect to group lutetium as an f-block element.[65] Since then, physical, chemical, and electronic evidence has overwhelmingly supported that the f-block contains the elements La–Yb and Ac–No,[64][62] as shown here and as supported by IUPAC reports dating from 1988 (when the 1–18 group numbers were recommended)[62] and 2021.[63] The variation nonetheless still exists because most textbook writers are not aware of the issue.[64] A third form can sometimes be encountered in which the spaces below yttrium in group 3 are left empty, but that contradicts quantum mechanics by making the f-block 15 elements wide (La–Lu and Ac–Lr) even though only 14 electrons can fit in an f-subshell.[63]
Some arguments in favour of Sc-Y-La-Ac can still be encountered in the literature, but many authors challenge them as being logically inconsistent.[43][39][66] For example, it has been argued that lanthanum and actinium cannot be f-block elements because their atoms have not begun to fill the f-subshells.[88] But the same is true of thorium which is never disputed as an f-block element,[63][64] and this argument overlooks the problem on the other end: that the f-shells complete filling at ytterbium and nobelium (matching the Sc-Y-Lu-Lr form), not at lutetium and lawrencium (as in Sc-Y-La-Ac).[89] Such exceptions to the Madelung rule have in any case never been considered as relevant for positioning any other elements on the periodic table.[68] The relevant fact for placement is that lanthanum and actinium (like thorium) have valence f-orbitals that can become occupied in chemical environments, whereas lutetium and lawrencium do not.[38][90] Thus the relationship between yttrium and lanthanum is only a secondary relationship between elements with the same number of valence electrons but different kinds of valence orbitals, such as that between chromium and uranium; whereas the relationship between yttrium and lutetium is primary, sharing both valence electron count and valence orbital type.[38]
Periodic trends
As chemical reactions involve the valence electrons,[5] elements with similar outer electron configurations may be expected to react similarly and form compounds with similar proportions of elements in them.[91] Such elements are placed in the same group, and thus there tend to be clear similarities and trends in chemical behaviour as one proceeds down a group.[92] As analogous configurations return at regular intervals, the properties of the elements thus exhibit periodic recurrences, hence the name of the periodic table and the periodic law. These periodic recurrences were noticed well before the underlying theory that explains them was developed.[93][94]
Atomic radius
Historically, the physical size of atoms was unknown until the early 20th century. The first calculated estimate of the atomic radius of hydrogen was published by physicist Artur Haas in 1910 to within an order of magnitude (a factor of 10) of the accepted value, the Bohr radius (~0.529 Å). In his model, Haas used a single-electron configuration based on the classical atomic model proposed by J. J. Thomson in 1904, often called the plum-pudding model.[95]
Atomic radii (the size of atoms) are dependent on the sizes of their outermost orbitals.[86] They generally decrease going left to right along the main-group elements, because the nuclear charge increases but the outer electrons are still in the same shell. However, going down a column, the radii generally increase, because the outermost electrons are in higher shells that are thus further away from the nucleus.[5][96] The first row of each block is abnormally small, due to an effect called kainosymmetry or primogenic repulsion:[97] the 1s, 2p, 3d, and 4f subshells have no inner analogues to which they would be orthogonal. Higher s-, p-, d-, and f-subshells experience strong repulsion from their inner analogues, which have approximately the same angular distribution of charge, and must expand to avoid this. This makes significant differences arise between the small 2p elements, which prefer multiple bonding, and the larger 3p and higher p-elements, which do not.[86] Similar anomalies arise for the 1s, 2p, 3d, 4f, and the hypothetical 5g elements:[98] the degree of this first-row anomaly is highest for the s-block, is moderate for the p-block, and is less pronounced for the d- and f-blocks.[99]
In the transition elements, an inner shell is filling, but the size of the atom is still determined by the outer electrons. The increasing nuclear charge across the series and the increased number of inner electrons for shielding somewhat compensate each other, so the decrease in radius is smaller.[96] The 4p and 5d atoms, coming immediately after new types of transition series are first introduced, are smaller than would have been expected,[100] because the added core 3d and 4f subshells provide only incomplete shielding of the nuclear charge for the outer electrons. Hence for example gallium atoms are slightly smaller than aluminium atoms.[86] Together with kainosymmetry, this results in an even-odd difference between the periods (except in the s-block)[i] that is sometimes known as secondary periodicity: elements in even periods have smaller atomic radii and prefer to lose fewer electrons, while elements in odd periods (except the first) differ in the opposite direction. Thus for example many properties in the p-block show a zigzag rather than a smooth trend along the group. For example, phosphorus and antimony in odd periods of group 15 readily reach the +5 oxidation state, whereas nitrogen, arsenic, and bismuth in even periods prefer to stay at +3.[99][101]
Thallium and lead atoms are about the same size as indium and tin atoms respectively, but from bismuth to radon the 6p atoms are larger than the analogous 5p atoms. This happens because when atomic nuclei become highly charged, special relativity becomes needed to gauge the effect of the nucleus on the electron cloud. These relativistic effects result in heavy elements increasingly having differing properties compared to their lighter homologues in the periodic table. Spin–orbit interaction splits the p-subshell: one p-orbital is relativistically stabilised and shrunken (it fills in thallium and lead), but the other two (filling in bismuth through radon) are relativistically destabilised and expanded.[86] Relativistic effects also explain why gold is golden and mercury is a liquid at room temperature.[102][103] They are expected to become very strong in the late seventh period, potentially leading to a collapse of periodicity.[104] Electron configurations are only clearly known until element 108 (hassium), and experimental chemistry beyond 108 has only been done for 112 (copernicium), 113 (nihonium), and 114 (flerovium), so the chemical characterisation of the heaviest elements remains a topic of current research.[105]
Ionisation energy
Graph of first ionisation energies of the elements in electronvolts (predictions used for elements 105–118)
The first ionisation energy of an atom is the energy required to remove an electron from it. This varies with the atomic radius: ionisation energy increases left to right and down to up, because electrons that are closer to the nucleus are held more tightly and are more difficult to remove. Ionisation energy thus is minimised at the first element of each period – hydrogen and the alkali metals – and then generally rises until it reaches the noble gas at the right edge of the period.[5] There are some exceptions to this trend, such as oxygen, where the electron being removed is paired and thus interelectronic repulsion makes it easier to remove than expected.[106]
In the transition series, the outer electrons are preferentially lost even though the inner orbitals are filling. For example, in the 3d series, the 4s electrons are lost first even though the 3d orbitals are being filled. The shielding effect of adding an extra 3d electron approximately compensates the rise in nuclear charge, and therefore the ionisation energies stay mostly constant, though there is a small increase especially at the end of each transition series.[107]
As metal atoms tend to lose electrons in chemical reactions, ionisation energy is generally correlated with chemical reactivity, although there are other factors involved as well.[107]
Electron affinity
Trend in electron affinities
The opposite property to ionisation energy is the electron affinity, which is the energy released when adding an electron to the atom.[108] A passing electron will be more readily attracted to an atom if it feels the pull of the nucleus more strongly, and especially if there is an available partially filled outer orbital that can accommodate it. Therefore, electron affinity tends to increase down to up and left to right. The exception is the last column, the noble gases, which have a full shell and have no room for another electron. This gives the halogens in the next-to-last column the highest electron affinities.[5]
Some atoms, like the noble gases, have no electron affinity: they cannot form stable gas-phase anions.[109] The noble gases, having high ionisation energies and no electron affinity, have little inclination towards gaining or losing electrons and are generally unreactive.[5]
Some exceptions to the trends occur: oxygen and fluorine have lower electron affinities than their heavier homologues sulfur and chlorine, because they are small atoms and hence the newly added electron would experience significant repulsion from the already present ones. For the nonmetallic elements, electron affinity likewise somewhat correlates with reactivity, but not perfectly since other factors are involved. For example, fluorine has a lower electron affinity than chlorine (because of extreme interelectronic repulsion for the very small fluorine atom), but is more reactive.[108]
Valence and oxidation states
The valence of an element can be defined either as the number of hydrogen atoms that can combine with it to form a simple binary hydride, or as twice the number of oxygen atoms that can combine with it to form a simple binary oxide (that is, not a peroxide or a superoxide). The valences of the main-group elements are directly related to the group number: the hydrides in the main groups 1–2 and 13–17 follow the formulae MH, MH2, MH3, MH4, MH3, MH2, and finally MH. The highest oxides instead increase in valence, following the formulae M2O, MO, M2O3, MO2, M2O5, MO3, M2O7.[j] Today the notion of valence has been extended by that of the oxidation state, which is the formal charge left on an element when all other elements in a compound have been removed as their ions.[91]
The electron configuration suggests a ready explanation from the number of electrons available for bonding,[91] although a full explanation requires considering the energy that would be released in forming compounds with different valences rather than simply considering electron configurations alone.[110] For example, magnesium forms Mg2+ rather than Mg+ cations when dissolved in water, because the latter would spontaneously disproportionate into Mg0 and Mg2+ cations. This is because the enthalpy of hydration (surrounding the cation with water molecules) increases in magnitude with the charge and radius of the ion. In Mg+, the outermost orbital (which determines ionic radius) is still 3s, so the hydration enthalpy is small and insufficient to compensate the energy required to remove the electron; but ionising again to Mg2+ uncovers the core 2p subshell, making the hydration enthalpy large enough to allow magnesium(II) compounds to form. For similar reasons, the common oxidation states of the heavier p-block elements (where the ns electrons become lower in energy than the np) tend to vary by steps of 2, because that is necessary to uncover an inner subshell and decrease the ionic radius (e.g. Tl+ uncovers 6s, and Tl3+ uncovers 5d, so once thallium loses two electrons it tends to lose the third one as well). Analogous arguments based on orbital hybridisation can be used for the less electronegative p-block elements.[111][k]
For transition metals, common oxidation states are nearly always at least +2 for similar reasons (uncovering the next subshell); this holds even for the metals with anomalous dx+1s1 or dx+2s0 configurations (except for silver), because repulsion between d-electrons means that the movement of the second electron from the s- to the d-subshell does not appreciably change its ionisation energy.[112] Because ionising the transition metals further does not uncover any new inner subshells, their oxidation states tend to vary by steps of 1 instead.[111] The lanthanides and late actinides generally show a stable +3 oxidation state, removing the outer s-electrons and then (usually) one electron from the (n−2)f-orbitals, that are similar in energy to ns.[113] The common and maximum oxidation states of the d- and f-block elements tend to depend on the ionisation energies. As the energy difference between the (n−1)d and ns orbitals rises along each transition series, it becomes less energetically favourable to ionise further electrons. Thus, the early transition metal groups tend to prefer higher oxidation states, but the +2 oxidation state becomes more stable for the late transition metal groups. The highest formal oxidation state thus increases from +3 at the beginning of each d-block row, to +7 or +8 in the middle (e.g. OsO4), and then to +2 at the end.[112] The lanthanides and late actinides usually have high fourth ionisation energies and hence rarely surpass the +3 oxidation state, whereas early actinides have low fourth ionisation energies and so for example neptunium and plutonium can reach +7.[112][113]
As elements in the same group share the same valence configurations, they usually exhibit similar chemical behaviour. For example, the alkali metals in the first group all have one valence electron, and form a very homogeneous class of elements: they are all soft and reactive metals. However, there are many factors involved, and groups can often be rather hetereogeneous. For instance, hydrogen also has one valence electron and is in the same group as the alkali metals, but its chemical behaviour is quite different. The stable elements of group 14 comprise a nonmetal (carbon), two semiconductors (silicon and germanium), and two metals (tin and lead); they are nonetheless united by having four valence electrons.[114] This often leads to similarities in maximum and minimum oxidation states (e.g. sulfur and selenium in group 16 both have maximum oxidation state +6, as in SO3 and SeO3, and minimum oxidation state −2, as in sulfides and selenides); but not always (e.g. oxygen is not known to form oxidation state +6, despite being in the same group as sulfur and selenium).[38]
Electronegativity
Another important property of elements is their electronegativity. Atoms can form covalent bonds to each other by sharing electrons in pairs, creating an overlap of valence orbitals. The degree to which each atom attracts the shared electron pair depends on the atom’s electronegativity[115] – the tendency of an atom towards gaining or losing electrons.[5] The more electronegative atom will tend to attract the electron pair more, and the less electronegative (or more electropositive) one will attract it less. In extreme cases, the electron can be thought of as having been passed completely from the more electropositive atom to the more electronegative one, though this is a simplification. The bond then binds two ions, one positive (having given up the electron) and one negative (having accepted it), and is termed an ionic bond.[5]
Electronegativity depends on how strongly the nucleus can attract an electron pair, and so it exhibits a similar variation to the other properties already discussed: electronegativity tends to fall going up to down, and rise going left to right. The alkali and alkaline earth metals are among the most electropositive elements, while the chalcogens, halogens, and noble gases are among the most electronegative ones.[115]
Electronegativity is generally measured on the Pauling scale, on which the most electronegative reactive atom (fluorine) is given electronegativity 4.0, and the least electronegative atom (caesium) is given electronegativity 0.79.[5] In fact neon is the most electronegative element, but the Pauling scale cannot measure its electronegativity because it does not form covalent bonds with most elements.[116]
An element’s electronegativity varies with the identity and number of the atoms it is bonded to, as well as how many electrons it has already lost: an atom becomes more electronegative when it has lost more electrons.[115] This sometimes makes a large difference: lead in the +2 oxidation state has electronegativity 1.87 on the Pauling scale, while lead in the +4 oxidation state has electronegativity 2.33.[117]
Metallicity
A simple substance is a substance formed from atoms of one chemical element. The simple substances of the more electronegative atoms tend to share electrons (form covalent bonds) with each other. They form either small molecules (like hydrogen or oxygen, whose atoms bond in pairs) or giant structures stretching indefinitely (like carbon or silicon). The noble gases simply stay as single atoms, as they already have a full shell.[5] Substances composed of discrete molecules or single atoms are held together by weaker attractive forces between the molecules, such as the London dispersion force: as electrons move within the molecules, they create momentary imbalances of electrical charge, which induce similar imbalances on nearby molecules and create synchronised movements of electrons across many neighbouring molecules.[118]
The more electropositive atoms, however, tend to instead lose electrons, creating a «sea» of electrons engulfing cations.[5] The outer orbitals of one atom overlap to share electrons with all its neighbours, creating a giant structure of molecular orbitals extending over all the atoms.[119] This negatively charged «sea» pulls on all the ions and keeps them together in a metallic bond. Elements forming such bonds are often called metals; those which do not are often called nonmetals.[5] Some elements can form multiple simple substances with different structures: these are called allotropes. For example, diamond and graphite are two allotropes of carbon.[114][l]
The metallicity of an element can be predicted from electronic properties. When atomic orbitals overlap during metallic or covalent bonding, they create both bonding and antibonding molecular orbitals of equal capacity, with the antibonding orbitals of higher energy. Net bonding character occurs when there are more electrons in the bonding orbitals than there are in the antibonding orbitals. Metallic bonding is thus possible when the number of electrons delocalised by each atom is less than twice the number of orbitals contributing to the overlap. This is the situation for elements in groups 1 through 13; they also have too few valence electrons to form giant covalent structures where all atoms take equivalent positions, and so almost all of them metallise. The exceptions are hydrogen and boron, which have too high an ionisation energy. Hydrogen thus forms a covalent H2 molecule, and boron forms a giant covalent structure based on icosahedral B12 clusters. In a metal, the bonding and antibonding orbitals have overlapping energies, creating a single band that electrons can freely flow through, allowing for electrical conduction.[121]
In group 14, both metallic and covalent bonding become possible. In a diamond crystal, covalent bonds between carbon atoms are strong, because they have a small atomic radius and thus the nucleus has more of a hold on the electrons. Therefore, the bonding orbitals that result are much lower in energy than the antibonding orbitals, and there is no overlap, so electrical conduction becomes impossible: carbon is a nonmetal. However, covalent bonding becomes weaker for larger atoms and the energy gap between the bonding and antibonding orbitals decreases. Therefore, silicon and germanium have smaller band gaps and are semiconductors: electrons can cross the gap when thermally excited. The band gap disappears in tin, so that tin and lead become metals.[121]
Elements in groups 15 through 17 have too many electrons to form giant covalent molecules that stretch in all three dimensions. For the lighter elements, the bonds in small diatomic molecules are so strong that a condensed phase is disfavoured: thus nitrogen (N2), oxygen (O2), white phosphorus and yellow arsenic (P4 and As4), sulfur and red selenium (S8 and Se8), and the stable halogens (F2, Cl2, Br2, and I2) readily form covalent molecules with few atoms. The heavier ones tend to form long chains (e.g. red phosphorus, grey selenium, tellurium) or layered structures (e.g. carbon as graphite, black phosphorus, grey arsenic, grey antimony, bismuth) that only extend in one or two rather than three dimensions. Both kinds of structures can be found as allotropes of phosphorus, arsenic, and selenium, although the long-chained allotropes are more stable in all three. As these structures do not use all their orbitals for bonding, they end up with bonding, nonbonding, and antibonding bands in order of increasing energy. Similarly to group 14, the band gaps shrink for the heavier elements and free movement of electrons between the chains or layers becomes possible. Thus for example black phosphorus, black arsenic, grey selenium, tellurium, and iodine are semiconductors; grey arsenic, grey antimony, and bismuth are semimetals (exhibiting quasi-metallic conduction, with a very small band overlap); and polonium and probably astatine are true metals.[121] Finally, the natural group 18 elements all stay as individual atoms.[121][m]
The dividing line between metals and nonmetals is roughly diagonal from top left to bottom right, with the transition series appearing to the left of this diagonal (as they have many available orbitals for overlap). This is expected, as metallicity tends to be correlated with electropositivity and the willingness to lose electrons, which increases right to left and up to down. Thus the metals greatly outnumber the nonmetals. Elements near the borderline are difficult to classify: they tend to have properties that are intermediate between those of metals and nonmetals, and may have some properties characteristic of both. They are often termed semimetals or metalloids.[5] The term «semimetal» used in this sense should not be confused with its strict physical sense having to do with band structure: bismuth is physically a semimetal, but is generally considered a metal by chemists.[123]
The following table considers the most stable allotropes at standard conditions. The elements coloured yellow form simple substances that are well-characterised by metallic bonding. Elements coloured light blue form giant network covalent structures, whereas those coloured dark blue form small covalently bonded molecules that are held together by weaker van der Waals forces. The noble gases are coloured in violet: their molecules are single atoms and no covalent bonding occurs. Greyed-out cells are for elements which have not been prepared in sufficient quantities for their most stable allotropes to have been characterised in this way. Theoretical considerations and current experimental evidence suggest that all of those elements would metallise if they could form condensed phases,[121] except perhaps for oganesson.[124][n]
Bonding of simple substances in the periodic table |
||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | |||||||||||||||
| Group → | ||||||||||||||||||||||||||||||||
| ↓ Period | ||||||||||||||||||||||||||||||||
| 1 | H | He | ||||||||||||||||||||||||||||||
| 2 | Li | Be | B | C | N | O | F | Ne | ||||||||||||||||||||||||
| 3 | Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||||||||||||||||
| 4 | K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | ||||||||||||||
| 5 | Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | ||||||||||||||
| 6 | Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| 7 | Fr | Ra | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
-
Iron, a metal
-
Sulfur, a nonmetal
-
Arsenic, an element often called a semi-metal or metalloid
Generally, metals are shiny and dense.[5] They usually have high melting and boiling points due to the strength of the metallic bond, and are often malleable and ductile (easily stretched and shaped) because the atoms can move relative to each other without breaking the metallic bond.[133] They conduct electricity because their electrons are free to move in all three dimensions. Similarly, they conduct heat, which is transferred by the electrons as extra kinetic energy: they move faster. These properties persist in the liquid state, as although the crystal structure is destroyed on melting, the atoms still touch and the metallic bond persists, though it is weakened.[133] Metals tend to be reactive towards nonmetals.[5] Some exceptions can be found to these generalisations: for example, manganese,[134] arsenic, antimony,[135] and bismuth are brittle;[136] chromium is extremely hard;[137] gallium, rubidium, caesium, and mercury are liquid at or close to room temperature;[o] and noble metals such as gold are chemically very inert.[138][139]
Nonmetals exhibit different properties. Those forming giant covalent crystals exhibit high melting and boiling points, as it takes considerable energy to overcome the strong covalent bonds. Those forming discrete molecules are held together mostly by dispersion forces, which are more easily overcome; thus they tend to have lower melting and boiling points,[140] and many are liquids or gases at room temperature.[5] Nonmetals are often dull-looking. They tend to be reactive towards metals, except for the noble gases, which are inert towards most substances.[5] They are brittle when solid as their atoms are held tightly in place. They are less dense and conduct electricity poorly,[5] because there are no mobile electrons.[141] Near the borderline, band gaps are small and thus many elements in that region are semiconductors, such as silicon, germanium,[141] selenium, and tellurium.[121] Again there are exceptions; for example, diamond has the highest thermal conductivity of all known materials, greater than any metal.[142]
It is common to designate a class of metalloids straddling the boundary between metals and nonmetals, as elements in that region are intermediate in both physical and chemical properties.[5] However, no consensus exists in the literature for precisely which elements should be so designated. When such a category is used, silicon, germanium, arsenic, and tellurium are almost always included, and boron and antimony usually are; but most sources include other elements as well, without agreement on which extra elements should be added, and some others subtract from this list instead.[p]
Further manifestations of periodicity
There are some other relationships throughout the periodic table between elements that are not in the same group, such as the diagonal relationships between elements that are diagonally adjacent (e.g. lithium and magnesium).[99] Some similarities can also be found between the main groups and the transition metal groups, or between the early actinides and early transition metals, when the elements have the same number of valence electrons. Thus uranium somewhat resembles chromium and tungsten in group 6,[99] as all three have six valence electrons.[147] Relationships between elements with the same number of valence electrons but different types of valence orbital have been called secondary or isodonor relationships: they usually have the same maximum oxidation states, but not the same minimum oxidation states. For example, chlorine and manganese both have +7 as their maximum oxidation state (e.g. Cl2O7 and Mn2O7), but their respective minimum oxidation states are −1 (e.g. HCl) and −3 (K2[Mn(CO)4]). Elements with the same number of valence vacancies but different numbers of valence electrons are related by a tertiary or isoacceptor relationship: they have similar minimum but not maximum oxidation states. For example, hydrogen and chlorine both have −1 as their minimum oxidation state (in hydrides and chlorides), but hydrogen’s maximum oxidation state is +1 (e.g. H2O) while chlorine’s is +7.[38]
Many other physical properties of the elements exhibit periodic variation in accordance with the periodic law, such as melting points, boiling points, heats of fusion, heats of vaporisation, atomisation energy, and so on. Similar periodic variations appear for the compounds of the elements, which can be observed by comparing hydrides, oxides, sulfides, halides, and so on.[115] Chemical properties are more difficult to describe quantitatively, but likewise exhibit their own periodicities. Examples include the variation in the acidic and basic properties of the elements and their compounds, the stabilities of compounds, and methods of isolating the elements.[91] Periodicity is and has been used very widely to predict the properties of unknown new elements and new compounds, and is central to modern chemistry.[148]
Classification of elements
A periodic table colour-coded to show some commonly used sets of similar elements. The categories and their boundaries differ somewhat between sources.[143] Lutetium and lawrencium in group 3 are also transition metals.[38]
Many terms have been used in the literature to describe sets of elements that behave similarly. The group names alkali metal, alkaline earth metal, triel, tetrel, pnictogen, chalcogen, halogen, and noble gas are acknowledged by IUPAC; the other groups can be referred to by their number, or by their first element (e.g., group 6 is the chromium group).[61][149] Some divide the p-block elements from groups 13 to 16 by metallicity,[145][143] although there is neither an IUPAC definition nor a precise consensus on exactly which elements should be considered metals, nonmetals, or semi-metals (sometimes called metalloids).[145][143][61] Neither is there a consensus on what the metals succeeding the transition metals ought to be called, with post-transition metal and poor metal being among the possibilities having been used.[q] Some advanced monographs exclude the elements of group 12 from the transition metals on the grounds of their sometimes quite different chemical properties, but this is not a universal practice[150] and IUPAC does not presently mention it as allowable in its Principles of Chemical Nomenclature.[151]
The lanthanides are considered to be the elements La–Lu, which are all very similar to each other: historically they included only Ce–Lu, but lanthanum became included by common usage.[61] The rare earth elements (or rare earth metals) add scandium and yttrium to the lanthanides.[61] Analogously, the actinides are considered to be the elements Ac–Lr (historically Th–Lr),[61] although variation of properties in this set is much greater than within the lanthanides.[35] IUPAC recommends the names lanthanoids and actinoids to avoid ambiguity, as the -ide suffix typically denotes a negative ion; however lanthanides and actinides remain common.[61] With the increasing recognition of lutetium and lawrencium as d-block elements, some authors began to define the lanthanides as La–Yb and the actinides as Ac–No, matching the f-block.[37][64][152][153][154]
Many more categorisations exist and are used according to certain disciplines. In astrophysics, a metal is defined as any element with atomic number greater than 2, i.e. anything except hydrogen and helium.[155] The term «semimetal» has a different definition in physics than it does in chemistry: bismuth is a semimetal by physical definitions, but chemists generally consider it a metal.[156] A few terms are widely used, but without any very formal definition, such as «heavy metal», which has been given such a wide range of definitions that it has been criticised as «effectively meaningless».[157]
The scope of terms varies significantly between authors. For example, according to IUPAC, the noble gases extend to include the whole group, including the very radioactive superheavy element oganesson.[158] However, among those who specialise in the superheavy elements, this is not often done: in this case «noble gas» is typically taken to imply the unreactive behaviour of the lighter elements of the group. Since calculations generally predict that oganesson should not be particularly inert due to relativistic effects, and may not even be a gas at room temperature if it could be produced in bulk, its status as a noble gas is often questioned in this context.[159] Furthermore, national variations are sometimes encountered: in Japan, alkaline earth metals often do not include beryllium and magnesium as their behaviour is different from the heavier group 2 metals.[160]
History
Mendeleev’s 1869 periodic table
Early history
In 1817, German physicist Johann Wolfgang Döbereiner began to formulate one of the earliest attempts to classify the elements.[161] In 1829, he found that he could form some of the elements into groups of three, with the members of each group having related properties. He termed these groups triads.[162][163] Chlorine, bromine, and iodine formed a triad; as did calcium, strontium, and barium; lithium, sodium, and potassium; and sulfur, selenium, and tellurium. Today, all these triads form part of modern-day groups.[164] Various chemists continued his work and were able to identify more and more relationships between small groups of elements. However, they could not build one scheme that encompassed them all.[165]
John Newlands published a letter in the Chemical News in February 1863 on the periodicity among the chemical elements.[166] In 1864 Newlands published an article in the Chemical News showing that if the elements are arranged in the order of their atomic weights, those having consecutive numbers frequently either belong to the same group or occupy similar positions in different groups, and he pointed out that each eighth element starting from a given one is in this arrangement a kind of repetition of the first, like the eighth note of an octave in music (The Law of Octaves).[166] However, Newlands’ formulation only worked well for the main-group elements, and encountered serious problems with the others.[38]
German chemist Lothar Meyer noted the sequences of similar chemical and physical properties repeated at periodic intervals. According to him, if the atomic weights were plotted as ordinates (i.e. vertically) and the atomic volumes as abscissas (i.e. horizontally)—the curve obtained a series of maximums and minimums—the most electropositive elements would appear at the peaks of the curve in the order of their atomic weights. In 1864, a book of his was published; it contained an early version of the periodic table containing 28 elements, and classified elements into six families by their valence—for the first time, elements had been grouped according to their valence. Works on organizing the elements by atomic weight had until then been stymied by inaccurate measurements of the atomic weights.[167] In 1868, he revised his table, but this revision was published as a draft only after his death.[168]
Mendeleev’s 1871 periodic table
Mendeleev
The definitive breakthrough came from the Russian chemist Dmitri Mendeleev. Although other chemists (including Meyer) had found some other versions of the periodic system at about the same time, Mendeleev was the most dedicated to developing and defending his system, and it was his system that most impacted the scientific community.[169] On 17 February 1869 (1 March 1869 in the Gregorian calendar), Mendeleev began arranging the elements and comparing them by their atomic weights. He began with a few elements, and over the course of the day his system grew until it encompassed most of the known elements. After he found a consistent arrangement, his printed table appeared in May 1869 in the journal of the Russian Chemical Society.[170] When elements did not appear to fit in the system, he boldly predicted that either valencies or atomic weights had been measured incorrectly, or that there was a missing element yet to be discovered.[38] In 1871, Mendeleev published a long article, including an updated form of his table, that made his predictions for unknown elements explicit. Mendeleev predicted the properties of three of these unknown elements in detail: as they would be missing heavier homologues of boron, aluminium, and silicon, he named them eka-boron, eka-aluminium, and eka-silicon («eka» being Sanskrit for «one»).[170][171]: 45
In 1875, the French chemist Paul-Émile Lecoq de Boisbaudran, working without knowledge of Mendeleev’s prediction, discovered a new element in a sample of the mineral sphalerite, and named it gallium. He isolated the element and began determining its properties. Mendeleev, reading de Boisbaudran’s publication, sent a letter claiming that gallium was his predicted eka-aluminium. Although Lecoq de Boisbaudran was initially sceptical, and suspected that Mendeleev was trying to take credit for his discovery, he later admitted that Mendeleev was correct.[172] In 1879, the Swedish chemist Lars Fredrik Nilson discovered a new element, which he named scandium: it turned out to be eka-boron. Eka-silicon was found in 1886 by German chemist Clemens Winkler, who named it germanium. The properties of gallium, scandium, and germanium matched what Mendeleev had predicted.[173] In 1889, Mendeleev noted at the Faraday Lecture to the Royal Institution in London that he had not expected to live long enough «to mention their discovery to the Chemical Society of Great Britain as a confirmation of the exactitude and generality of the periodic law».[174] Even the discovery of the noble gases at the close of the 19th century, which Mendeleev had not predicted, fitted neatly into his scheme as an eighth main group.[175]
Mendeleev nevertheless had some trouble fitting the known lanthanides into his scheme, as they did not exhibit the periodic change in valencies that the other elements did. After much investigation, the Czech chemist Bohuslav Brauner suggested in 1902 that the lanthanides could all be placed together in one group on the periodic table. He named this the «asteroid hypothesis» as an astronomical analogy: just as there is an asteroid belt instead of a single planet between Mars and Jupiter, so the place below yttrium was thought to be occupied by all the lanthanides instead of just one element.[68]
Atomic number
After the internal structure of the atom was probed, amateur Dutch physicist Antonius van den Broek proposed in 1913 that the nuclear charge determined the placement of elements in the periodic table.[176][177] The New Zealand physicist Ernest Rutherford coined the word «atomic number» for this nuclear charge.[178] In van der Broek’s published article he illustrated the first electronic periodic table showing the elements arranged according to the number of their electrons.[179] Rutherford confirmed in his 1914 paper that Bohr had accepted the view of van der Broek.[180]
Periodic table of van den Broek
The same year, English physicist Henry Moseley using X-ray spectroscopy confirmed van den Broek’s proposal experimentally. Moseley determined the value of the nuclear charge of each element from aluminium to gold and showed that Mendeleev’s ordering actually places the elements in sequential order by nuclear charge.[181] Nuclear charge is identical to proton count and determines the value of the atomic number (Z) of each element. Using atomic number gives a definitive, integer-based sequence for the elements. Moseley’s research immediately resolved discrepancies between atomic weight and chemical properties; these were cases such as tellurium and iodine, where atomic number increases but atomic weight decreases.[176] Although Moseley was soon killed in World War I, the Swedish physicist Manne Siegbahn continued his work up to uranium, and established that it was the element with the highest atomic number then known (92).[182] Based on Moseley and Siegbahn’s research, it was also known which atomic numbers corresponded to missing elements yet to be found.[176]
Electron shells
The Danish physicist Niels Bohr applied Max Planck’s idea of quantisation to the atom. He concluded that the energy levels of electrons were quantised: only a discrete set of stable energy states were allowed. Bohr then attempted to understand periodicity through electron configurations, surmising in 1913 that the inner electrons should be responsible for the chemical properties of the element.[183][184] In 1913, he produced the first electronic periodic table based on a quantum atom.[185]
Bohr called his electron shells «rings» in 1913: atomic orbitals within shells did not exist at the time of his planetary model. Bohr explains in Part 3 of his famous 1913 paper that the maximum electrons in a shell is eight, writing, «We see, further, that a ring of n electrons cannot rotate in a single ring round a nucleus of charge ne unless n < 8.» For smaller atoms, the electron shells would be filled as follows: «rings of electrons will only join if they contain equal numbers of electrons; and that accordingly the numbers of electrons on inner rings will only be 2, 4, 8.» However, in larger atoms the innermost shell would contain eight electrons: «on the other hand, the periodic system of the elements strongly suggests that already in neon N = 10 an inner ring of eight electrons will occur.» His proposed electron configurations for the atoms (shown to the right) mostly do not accord with those now known.[186][187]
| Element | Electrons per shell |
|---|---|
| 4 | 2,2 |
| 6 | 2,4 |
| 7 | 4,3 |
| 8 | 4,2,2 |
| 9 | 4,4,1 |
| 10 | 8,2 |
| 11 | 8,2,1 |
| 16 | 8,4,2,2 |
| 18 | 8,8,2 |
The first one to systematically expand and correct the chemical potentials of Bohr’s atomic theory was Walther Kossel in 1914 and in 1916. Kossel explained that in the periodic table new elements would be created as electrons were added to the outer shell. In Kossel’s paper, he writes: «This leads to the conclusion that the electrons, which are added further, should be put into concentric rings or shells, on each of which … only a certain number of electrons—namely, eight in our case—should be arranged. As soon as one ring or shell is completed, a new one has to be started for the next element; the number of electrons, which are most easily accessible, and lie at the outermost periphery, increases again from element to element and, therefore, in the formation of each new shell the chemical periodicity is repeated.»[188][189]
Periodic table of Alfred Werner (1905), the first appearance of the long form[68]
In a 1919 paper, Irving Langmuir postulated the existence of «cells» which we now call orbitals, which could each only contain two electrons each, and these were arranged in «equidistant layers» which we now call shells. He made an exception for the first shell to only contain two electrons.[190] The chemist Charles Rugeley Bury suggested in 1921 that eight and eighteen electrons in a shell form stable configurations. Bury proposed that the electron configurations in transitional elements depended upon the valence electrons in their outer shell.[191] He introduced the word transition to describe the elements now known as transition metals or transition elements.[192]
Prompted by Bohr, Wolfgang Pauli took up the problem of electron configurations in 1923. Pauli extended Bohr’s scheme to use four quantum numbers, and formulated his exclusion principle which stated that no two electrons could have the same four quantum numbers. This explained the lengths of the periods in the periodic table (2, 8, 18, and 32), which corresponded to the number of electrons that each shell could occupy.[193] In 1925, Friedrich Hund arrived at configurations close to the modern ones.[194] As a result of these advances, periodicity became based on the number of chemically active or valence electrons rather than by the valences of the elements.[38] The Aufbau principle that describes the electron configurations of the elements was first empirically observed by Erwin Madelung in 1926,[28] though the first to publish it was Vladimir Karapetoff in 1930.[195][196] In 1961, Vsevolod Klechkovsky derived the first part of the Madelung rule (that orbitals fill in order of increasing n + ℓ) from the Thomas–Fermi model;[197] the complete rule was derived from a similar potential in 1971 by Yury N. Demkov and Valentin N. Ostrovsky.[198][r]
The quantum theory clarified the transition metals and lanthanides as forming their own separate groups, transitional between the main groups, although some chemists had already proposed tables showing them this way before then: the English chemist Henry Bassett did so in 1892, the Danish chemist Julius Thomsen in 1895, and the Swiss chemist Alfred Werner in 1905. Bohr used Thomsen’s form in his 1922 Nobel Lecture; Werner’s form is very similar to the modern 32-column form. In particular, this supplanted Brauner’s asteroidal hypothesis.[68]
The exact position of the lanthanides, and thus the composition of group 3, remained under dispute for decades longer because their electron configurations were initially measured incorrectly.[64][82] On chemical grounds Bassett, Werner, and Bury grouped scandium and yttrium with lutetium rather than lanthanum (the former two left an empty space below yttrium as lutetium had not yet been discovered).[68][191] Hund assumed in 1927 that all the lanthanide atoms had configuration [Xe]4f0−145d16s2, on account of their prevailing trivalency (it is now known that the relationship between chemistry and electron configuration is more complicated than that).[199] Early spectroscopic evidence seemed to confirm this, and thus the periodic table was structured to have group 3 as scandium, yttrium, lanthanum, and actinium, with fourteen f-elements breaking up the d-block between lanthanum and hafnium.[64] But it was later discovered that this is only true for four of the fifteen lanthanides (lanthanum, cerium, gadolinium, and lutetium), and that the other lanthanide atoms do not have a d-electron. In particular, ytterbium completes the 4f shell and thus Soviet physicists Lev Landau and Evgeny Lifshitz noted in 1948 that lutetium is correctly regarded as a d-block rather than an f-block element;[65] that bulk lanthanum is an f-metal was first suggested by Jun Kondō in 1963, on the grounds of its low-temperature superconductivity.[200] This clarified the importance of looking at low-lying excited states of atoms that can play a role in chemical environments when classifying elements by block and positioning them on the table.[44][45][64] Many authors subsequently rediscovered this correction based on physical, chemical, and electronic concerns and applied it to all the relevant elements, thus making group 3 contain scandium, yttrium, lutetium, and lawrencium[44][62][82] and having lanthanum through ytterbium and actinium through nobelium as the f-block rows:[44][62] this corrected version achieves consistency with the Madelung rule and vindicates Bassett, Werner, and Bury’s initial chemical placement.[68]
In 1988, IUPAC released a report supporting this composition of group 3,[62] a decision that was reaffirmed in 2021.[63] Variation can still be found in textbooks on the composition of group 3,[70] and some argumentation against this format is still published today,[43] but chemists and physicists who have considered the matter largely agree on group 3 containing scandium, yttrium, lutetium, and lawrencium and challenge the counterarguments as being inconsistent.[43]
Synthetic elements
By 1936, the pool of missing elements from hydrogen to uranium had shrunk to four: elements 43, 61, 85, and 87 remained missing. Element 43 eventually became the first element to be synthesised artificially via nuclear reactions rather than discovered in nature. It was discovered in 1937 by Italian chemists Emilio Segrè and Carlo Perrier, who named their discovery technetium, after the Greek word for «artificial».[201] Elements 61 (promethium) and 85 (astatine) were likewise produced artificially in 1945 and 1940 respectively; element 87 (francium) became the last element to be discovered in nature, by French chemist Marguerite Perey in 1939.[202][s] The elements beyond uranium were likewise discovered artificially, starting with Edwin McMillan and Philip Abelson’s 1940 discovery of neptunium (via bombardment of uranium with neutrons).[55] Glenn T. Seaborg and his team at the Lawrence Berkeley National Laboratory (LBNL) continued discovering transuranium elements, starting with plutonium in 1941, and discovered that contrary to previous thinking, the elements from actinium onwards were congeners of the lanthanides rather than transition metals.[203] Bassett (1892), Werner (1905), and the French engineer Charles Janet (1928) had previously suggested this, but their ideas did not then receive general acceptance.[68] Seaborg thus called them the actinides.[203] Elements up to 101 (named mendelevium in honour of Mendeleev) were synthesised up to 1955, either through neutron or alpha-particle irradiation, or in nuclear explosions in the cases of 99 (einsteinium) and 100 (fermium).[55]
A significant controversy arose with elements 102 through 106 in the 1960s and 1970s, as competition arose between the LBNL team (now led by Albert Ghiorso) and a team of Soviet scientists at the Joint Institute for Nuclear Research (JINR) led by Georgy Flyorov. Each team claimed discovery, and in some cases each proposed their own name for the element, creating an element naming controversy that lasted decades. These elements were made by bombardment of actinides with light ions.[204] IUPAC at first adopted a hands-off approach, preferring to wait and see if a consensus would be forthcoming. Unfortunately, it was also the height of the Cold War, and it became clear after some time that this would not happen. As such, IUPAC and the International Union of Pure and Applied Physics (IUPAP) created a Transfermium Working Group (TWG, fermium being element 100) in 1985 to set out criteria for discovery,[205] which were published in 1991.[206] After some further controversy, these elements received their final names in 1997, including seaborgium (106) in honour of Seaborg.[207]
The TWG’s criteria were used to arbitrate later element discovery claims from LBNL and JINR, as well as from research institutes in Germany (GSI) and Japan (Riken).[208] Currently, consideration of discovery claims is performed by a IUPAC/IUPAP Joint Working Party. After priority was assigned, the elements were officially added to the periodic table, and the discoverers were invited to propose their names.[8] By 2016, this had occurred for all elements up to 118, therefore completing the periodic table’s first seven rows.[8][209] The discoveries of elements beyond 106 were made possible by techniques devised by Yuri Oganessian at the JINR: cold fusion (bombardment of lead and bismuth by heavy ions) made possible the 1981–2004 discoveries of elements 107 through 112 at GSI and 113 at Riken, and he led the JINR team (in collaboration with American scientists) to discover elements 114 through 118 using hot fusion (bombardment of actinides by calcium ions) in 1998–2010.[210][211] The heaviest known element, oganesson (118), is named in Oganessian’s honour. Element 114 is named flerovium in honour of his predecessor and mentor Flyorov.[211]
In celebration of the periodic table’s 150th anniversary, the United Nations declared the year 2019 as the International Year of the Periodic Table, celebrating «one of the most significant achievements in science».[212] The discovery criteria set down by the TWG were updated in 2020 in response to experimental and theoretical progress that had not been foreseen in 1991.[213] Today, the periodic table is among the most recognisable icons of chemistry.[71] IUPAC is involved today with many processes relating to the periodic table: the recognition and naming of new elements, recommending group numbers and collective names, and the updating of atomic weights.[8]
Future extension beyond the seventh period
The most recently named elements – nihonium (113), moscovium (115), tennessine (117), and oganesson (118) – completed the seventh row of the periodic table.[8] Future elements would have to begin an eighth row. These elements may be referred to either by their atomic numbers (e.g. «element 119»), or by the IUPAC systematic element names adopted in 1978, which directly relate to the atomic numbers (e.g. «ununennium» for element 119, derived from Latin unus «one», Greek ennea «nine», and the traditional -ium suffix for metallic elements).[8] All attempts to synthesise such elements have failed so far. An attempt to make element 119 has been ongoing since 2018 at the Riken research institute in Japan. The Joint Institute for Nuclear Research in Russia also plans to make its own attempts at synthesising the first few period 8 elements.[214][215][216]
If the eighth period follows the pattern set by the earlier periods, then it would contain fifty elements, filling the 8s, 5g, 6f, 7d, and finally 8p subshells in that order. But there is present discussion regarding whether this would truly be the case, as calculations predict that by this point relativistic effects should result in significant deviations from the Madelung rule. Various different models have been suggested. All agree that the eighth period should begin like the previous ones with two 8s elements, and that there should then follow a new series of g-block elements filling up the 5g orbitals, but the precise configurations calculated for these 5g elements vary widely between sources. Beyond this 5g series, calculations do not agree on what exactly should follow. Filling of the 5g, 6f, 7d, and 8p shells is indeed expected to occur in approximately that order, but they are likely to be intermingled with each other and with the 9s and 9p subshells, so that it is not clear which elements should go in which groups anymore.[217][218][219][40][220] Eric Scerri has raised the question of whether an extended periodic table should take into account the failure of the Madelung rule in this region, or if such exceptions should be ignored.[220] The shell structure may also be fairly formal at this point: already the electron distribution in an oganesson atom is expected to be rather uniform, with no discernible shell structure.[221]
Nuclear stability will likely prove a decisive factor constraining the number of possible elements.[t] It depends on the balance between the electric repulsion between protons and the strong force binding protons and neutrons together.[224] Protons and neutrons are arranged in shells, just like electrons, and so a closed shell can significantly increase stability: the known superheavy nuclei exist because of such a shell closure. They are probably close to a predicted island of stability, where superheavy nuclides should have significantly longer half-lives: predictions range from minutes or days, to millions or billions of years.[225][226] However, as the number of protons increases beyond about 126, this stabilising effect should vanish as a closed shell is passed. It is not clear if any further-out shell closures exist, due to an expected smearing out of distinct nuclear shells (as is already expected for the electron shells at oganesson).[227] Furthermore, even if later shell closures exist, it is not clear if they would allow such heavy elements to exist.[228][229][230][145] As such, it may be that the periodic table practically ends around element 120, as elements become too short-lived to observe; the era of discovering new elements would thus be close to its end.[145][231]
Alternatively, quark matter may become stable at high mass numbers, in which the nucleus is composed of freely flowing up and down quarks instead of binding them into protons and neutrons; this would create a continent of stability instead of an island.[232][233] Other effects may come into play: for example, in very heavy elements the 1s electrons are likely to spend a significant amount of time so close to the nucleus that they are actually inside it, which would make them vulnerable to electron capture.[234]
Even if eighth-row elements can exist, producing them is likely to be difficult, and it should become even more difficult as atomic number rises.[235] Although the 8s elements are expected to be reachable with present means, the first few 5g elements are expected to require new technology,[236] if they can be produced at all.[237] Experimentally characterising these elements chemically would also pose a great challenge.[214]
Alternative periodic tables
The periodic law may be represented in multiple ways, of which the standard periodic table is only one.[238] Within 100 years of the appearance of Mendeleev’s table in 1869, Edward G. Mazurs had collected an estimated 700 different published versions of the periodic table.[147][239] Many forms retain the rectangular structure, including Charles Janet’s left-step periodic table (pictured below), and the modernised form of Mendeleev’s original 8-column layout that is still common in Russia. Other periodic table formats have been shaped much more exotically, such as spirals (Otto Theodor Benfey’s pictured to the right), circles and triangles.[240]
Alternative periodic tables are often developed to highlight or emphasize chemical or physical properties of the elements that are not as apparent in traditional periodic tables, with different ones skewed more towards emphasizing chemistry or physics at either end.[241] The standard form, which remains by far the most common, is somewhere in the middle.[241]
The many different forms of the periodic table have prompted the questions of whether there is an optimal or definitive form of the periodic table, and if so, what it might be. There are no current consensus answers to either question.[242][241] Janet’s left-step table is being increasingly discussed as a candidate for being the optimal or most fundamental form; Scerri has written in support of it, as it clarifies helium’s nature as an s-block element, increases regularity by having all period lengths repeated, faithfully follows Madelung’s rule by making each period correspond to one value of n + ℓ, and regularises atomic number triads and the first-row anomaly trend. While he notes that its placement of helium atop the alkaline earth metals can be seen a disadvantage from a chemical perspective, he counters this by appealing to the first-row anomaly, pointing out that the periodic table «fundamentally reduces to quantum mechanics», and that it is concerned with «abstract elements» and hence atomic properties rather than macroscopic properties.[243]
Notes
- ^ The question of how many natural elements there are is quite complicated and is not fully resolved. In the early Solar System, shorter-lived elements had not yet decayed away, and consequently there were more than 94 naturally occurring elements. Curium (element 96) is the longest-lived element beyond the first 94, and is probably still being brought to Earth via cosmic rays, but it has not been found.[10] Elements up to 99 (einsteinium) have been observed in Przybylski’s Star.[11] Elements up to 100 (fermium) probably occurred in the natural nuclear fission reactor at Oklo Mine, Gabon, but they have long since decayed away.[12] Even heavier elements may be produced in the r-process via supernovae or neutron star mergers, but this has not been confirmed. It is not clear how far they would extend past 100 and how long they would last: calculations suggest that nuclides of mass number around 280 to 290 are formed in the r-process, but quickly beta decay to nuclides that suffer spontaneous fission, so that 99.9% of the produced superheavy nuclides would decay within a month.[13] If instead they were sufficiently long-lived, they might similarly be brought to Earth via cosmic rays, but again none have been found.[10]
- ^ The half-life of plutonium’s most stable isotope is just long enough that it should also be a primordial element. A 1971 study claimed to have detected primordial plutonium,[15] but a more recent study from 2012 could not detect it.[16]
- ^ Tiny traces of plutonium are also continually brought to Earth via cosmic rays.[17]
- ^ Strictly speaking, one cannot draw an orbital such that the electron is guaranteed to be inside it, but it can be drawn to guarantee a 90% probability of this for example.[23]
- ^
Once two to four electrons are removed, the d and f orbitals usually become lower in energy than the s ones:[35]- 1s ≪ 2s < 2p ≪ 3s < 3p ≪ 3d < 4s < 4p ≪ 4d < 5s < 5p ≪ 4f < 5d < 6s < 6p ≪ 5f < 6d < 7s < 7p ≪ …
and in the limit for extremely highly charged ions, orbitals simply fill in the order of increasing n instead. There is a gradual transition between the limiting situations of highly charged ions (increasing n) and neutral atoms (Madelung’s rule).[28]
Also, the ordering of the orbitals between each ≪ changes somewhat throughout each period. For example, the ordering in argon and potassium is 3p ≪ 4s < 4p ≪ 3d; by calcium it has become 3p ≪ 4s < 3d < 4p; from scandium to copper it is 3p ≪ 3d < 4s < 4p; and from zinc to krypton it is 3p < 3d ≪ 4s < 4p[34] as the d-orbitals fall into the core at gallium.[36][37]
- ^ In fact, electron configurations represent a first-order approximation: an atom really exists in a superposition of multiple configurations, and electrons in an atom are indistinguishable.[39] The elements in the d- and f-blocks have multiple configurations separated by small energies and can change configuration depending on the chemical environment.[35] In some of the undiscovered g-block elements, it is predicted that mixing of configurations should become so important that the result can no longer be well-described by a single configuration.[40]
- ^ Compounds that would use the 6d orbitals of nihonium as valence orbitals have been theoretically investigated, but they are all expected to be too unstable to observe.[51]
- ^ See for example the periodic table poster sold by Sigma-Aldrich.
- ^ Properties of the p-block elements nevertheless do affect the succeeding s-block elements. The 3s shell in sodium is above a kainosymmetric 2p core, but the 4s shell in potassium is above the much larger 3p core. Hence while one would have already expected potassium atoms to be larger than sodium atoms, the size difference is greater than usual.[86]
- ^ There are many lower oxides as well: for example, phosphorus in group 15 forms two oxides, P2O3 and P2O5.[91]
- ^ The normally «forbidden» intermediate oxidation states may be stabilised by forming dimers, as in [Cl3Ga–GaCl3]2− (gallium in the +2 oxidation state) or S2F10 (sulfur in the +5 oxidation state).[111]
- ^ The boundary between dispersion forces and metallic bonding is gradual, like that between ionic and covalent bonding. Characteristic metallic properties do not appear in small mercury clusters, but do appear in large ones.[120]
- ^ All this describes the situation at standard pressure. Under sufficiently high pressure, the band gaps of any solid drop to zero and metallisation occurs. Thus for example at about 170 kbar iodine becomes a metal,[121] and metallic hydrogen should form at pressures of about four million atmospheres.[122]
- ^ Descriptions of the structures formed by the elements can be found throughout Greenwood and Earnshaw. There are two borderline cases. Arsenic’s most stable form conducts electricity like a metal, but the bonding is significantly more localised to the nearest neighbours than it is for the similar structures of antimony and bismuth.[125] Carbon as graphite shows metallic conduction parallel to its planes, but is a semiconductor perpendicular to them. Some computations predict copernicium and flerovium to be nonmetallic,[126][127] but the most recent experiments on them suggest that they are metallic.[128][129][130] Astatine is calculated to metallise at standard conditions,[131] so presumably tennessine should as well.[132]
- ^ See melting points of the elements (data page). The same is probably true of francium, but due to its extreme instability, this has never been experimentally confirmed. Copernicium and flerovium are expected to be liquids,[126][127] similar to mercury, and experimental evidence suggests that they are metals.[128][129][130]
- ^ See lists of metalloids. For example, a periodic table used by the American Chemical Society includes polonium as a metalloid,[143] but one used by the Royal Society of Chemistry does not,[144] and that included in the Encyclopædia Britannica does not refer to metalloids or semi-metals at all.[145] Classification can change even within a single work. For example, Sherwin and Weston’s Chemistry of the Non-Metallic Elements (1966) has a periodic table on p. 7 classifying antimony as a nonmetal, but on p. 115 it is called a metal.[146]
- ^ See post-transition metal.
- ^ Demkov and Ostrovsky consider the potential
where
and
are constant parameters; this approaches a Coulomb potential for small
. When
satisfies the condition
, where
, the zero-energy solutions to the Schrödinger equation for this potential can be described analytically with Gegenbauer polynomials. As
passes through each of these values, a manifold containing all states with that value of
arises at zero energy and then becomes bound, recovering the Madelung order. Perturbation-theory considerations show that states with smaller
have lower energy, and that the s-orbitals (with
) have their energies approaching the next
group.[198][81]
- ^ Technetium, promethium, astatine, neptunium, and plutonium were eventually discovered to occur in nature as well, albeit in tiny traces. See timeline of chemical element discoveries.
- ^ A simplistic interpretation of the relativistic Dirac equation runs into problems with electron orbitals at Z > 1/α ≈ 137; this would suggest that neutral atoms cannot exist beyond element 137, and that a periodic table of elements based on electron orbitals therefore breaks down at this point.[222] However, this argument presumes that the atomic nucleus is pointlike. A more accurate calculation must take into account the small, but nonzero, size of the nucleus, which pushes the limit to Z = 173. Moreover, it turns out that the prohibition is not against neutral atoms, but against bare nuclei: atoms with over 173 protons cannot be totally ionised because their 1s shell would be filled by spontaneous electron–positron pair production, but encounter no difficulties if their 1s shell is already filled.[223]
References
- ^ Meija, Juris; et al. (2016). «Atomic weights of the elements 2013 (IUPAC Technical Report)». Pure and Applied Chemistry. 88 (3): 265–91. doi:10.1515/pac-2015-0305.
- ^ Prohaska, Thomas; Irrgeher, Johanna; Benefield, Jacqueline; et al. (4 May 2022). «Standard atomic weights of the elements 2021 (IUPAC Technical Report)». Pure and Applied Chemistry. doi:10.1515/pac-2019-0603. ISSN 1365-3075.
- ^ Feynman, Richard; Leighton, Robert B.; Sands, Matthew (1964). «2. The Relation of Wave and Particle Viewpoints». The Feynman Lectures on Physics. Vol. 3. Addison–Wesley. ISBN 0-201-02115-3. Archived from the original on 19 October 2021. Retrieved 15 August 2021.
- ^ a b c Feynman, Richard; Leighton, Robert B.; Sands, Matthew (1964). «2. Basic Physics». The Feynman Lectures on Physics. Vol. 1. Addison–Wesley. ISBN 0-201-02115-3. Archived from the original on 28 March 2014. Retrieved 15 August 2021.
- ^ a b c d e f g h i j k l m n o p q r s t u Gonick, First; Criddle, Craig (2005). The Cartoon Guide to Chemistry. Collins. pp. 17–65. ISBN 0-06-093677-0.
- ^ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the «Gold Book») (1997). Online corrected version: (2006–) «Atomic number». doi:10.1351/goldbook.A00499
- ^ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the «Gold Book») (1997). Online corrected version: (2006–) «Chemical element». doi:10.1351/goldbook.C01022
- ^ a b c d e f g h i j k l m «Periodic Table of Elements». iupac.org. IUPAC. 2021. Archived from the original on 10 April 2016. Retrieved 3 April 2021.
- ^ Commission on Isotopic Abundances and Atomic Weights (2019). «Standard Atomic Weights». www.ciaaw.org. International Union of Pure and Applied Chemistry. Archived from the original on 8 August 2020. Retrieved 7 February 2021.
{{cite web}}: CS1 maint: uses authors parameter (link) - ^ a b c Thornton, Brett F.; Burdette, Shawn C. (2019). «Neutron stardust and the elements of Earth». Nature Chemistry. 11 (1): 4–10. Bibcode:2019NatCh..11….4T. doi:10.1038/s41557-018-0190-9. PMID 30552435. S2CID 54632815. Archived from the original on 14 August 2021. Retrieved 19 February 2022.
- ^ Gopka, V.F.; Yushchenko, A.V.; Yushchenko, V.A.; Panov, I.V.; Kim, Ch. (15 May 2008). «Identification of absorption lines of short half-life actinides in the spectrum of Przybylski’s star (HD 101065)». Kinematics and Physics of Celestial Bodies. 24 (2): 89–98. Bibcode:2008KPCB…24…89G. doi:10.3103/S0884591308020049. S2CID 120526363.
- ^ Emsley, John (2011). Nature’s Building Blocks: An A-Z guide to the elements (New ed.). New York, NY: Oxford University Press. ISBN 978-0-19-960563-7.
- ^ Panov, I.V. (2017). «Formation of Superheavy Elements in Nature». Physics of Atomic Nuclei. 81 (1): 57–65. doi:10.1134/S1063778818010167. S2CID 125149409.
- ^ Marcillac, Pierre de; Noël Coron; Gérard Dambier; Jacques Leblanc; Jean-Pierre Moalic (April 2003). «Experimental detection of α-particles from the radioactive decay of natural bismuth». Nature. 422 (6934): 876–878. Bibcode:2003Natur.422..876D. doi:10.1038/nature01541. PMID 12712201. S2CID 4415582.
- ^ Hoffman, D. C.; Lawrence, F. O.; Mewherter, J. L.; Rourke, F. M. (1971). «Detection of Plutonium-244 in Nature». Nature. 234 (5325): 132–134. Bibcode:1971Natur.234..132H. doi:10.1038/234132a0. S2CID 4283169.
- ^ Lachner, J.; et al. (2012). «Attempt to detect primordial 244Pu on Earth». Physical Review C. 85 (1): 015801. Bibcode:2012PhRvC..85a5801L. doi:10.1103/PhysRevC.85.015801.
- ^ Wallner, A.; Faestermann, T.; Feige, J.; Feldstein, C.; Knie, K.; Korschinek, G.; et al. (2015). «Abundance of live 244Pu in deep-sea reservoirs on Earth points to rarity of actinide nucleosynthesis». Nature Communications. 6: 5956. arXiv:1509.08054. Bibcode:2015NatCo…6.5956W. doi:10.1038/ncomms6956. ISSN 2041-1723. PMC 4309418. PMID 25601158.
- ^ a b Scerri, p. 17
- ^ «periodic law». Merriam-Webster Dictionary. Retrieved 29 March 2021.
- ^ a b Jensen, William B. (2009). «Misapplying the Periodic Law». Journal of Chemical Education. 86 (10): 1186. Bibcode:2009JChEd..86.1186J. doi:10.1021/ed086p1186.
- ^ a b c d e f g h i j k Feynman, Richard; Leighton, Robert B.; Sands, Matthew (1964). «19. The Hydrogen Atom and The Periodic Table». The Feynman Lectures on Physics. Vol. 3. Addison–Wesley. ISBN 0-201-02115-3. Archived from the original on 19 October 2021. Retrieved 15 August 2021.
- ^ Petrucci et al., p. 323
- ^ Petrucci et al., p. 306
- ^ Petrucci et al., p. 322
- ^ Ball, David W.; Key, Jessie A. (2011). Introductory Chemistry (1st Canadian ed.). Vancouver, British Columbia: BC Campus (opentextbc.ca). ISBN 978-1-77420-003-2. Archived from the original on 15 August 2021. Retrieved 15 August 2021.
- ^ «Electron Configurations». www.chem.fsu.edu. Florida State University. 6 May 2020. Archived from the original on 6 May 2022. Retrieved 17 April 2022.
- ^ a b c d e f g h i j Petrucci et al., p. 331
- ^ a b c d Goudsmit, S. A.; Richards, Paul I. (1964). «The Order of Electron Shells in Ionized Atoms» (PDF). Proc. Natl. Acad. Sci. 51 (4): 664–671 (with correction on p 906). Bibcode:1964PNAS…51..664G. doi:10.1073/pnas.51.4.664. PMC 300183. PMID 16591167. Archived (PDF) from the original on 10 October 2017. Retrieved 15 August 2021.
- ^ Jolly, William L. (1984). Modern Inorganic Chemistry (1st ed.). McGraw-Hill. pp. 10–12. ISBN 0-07-032760-2.
- ^ a b c d e Ostrovsky, V. N. (May 2001). «What and How Physics Contributes to Understanding the Periodic Law». Foundations of Chemistry. 3 (2): 145–181. doi:10.1023/A:1011476405933. S2CID 15679915.
- ^ a b Ostrovsky, V. N. (1981). «Dynamic symmetry of atomic potential». Journal of Physics B: Atomic and Molecular Physics. 14 (23): 4425–4439. Bibcode:1981JPhB…14.4425O. doi:10.1088/0022-3700/14/23/008.
- ^ a b Wong, D. Pan (1979). «Theoretical justification of Madelung’s rule». J. Chem. Educ. 56 (11): 714–718. Bibcode:1979JChEd..56..714W. doi:10.1021/ed056p714.
- ^ a b c Petrucci et al., p. 328
- ^ a b Cao, Changsu; Vernon, René E.; Schwarz, W. H. Eugen; Li, Jun (6 January 2021). «Understanding Periodic and Non-periodic Chemistry in Periodic Tables». Frontiers in Chemistry. 8 (813): 813. Bibcode:2021FrCh….8..813S. doi:10.3389/fchem.2020.00813. PMC 7818537. PMID 33490030.
- ^ a b c d e f g Jørgensen, Christian (1973). «The Loose Connection between Electron Configuration and the Chemical Behavior of the Heavy Elements (Transuranics)». Angewandte Chemie International Edition. 12 (1): 12–19. doi:10.1002/anie.197300121.
- ^ Tossell, J.A. (1 November 1977). «Theoretical studies of valence orbital binding energies in solid zinc sulfide, zinc oxide, and zinc fluoride». Inorganic Chemistry. 16 (11): 2944–2949. doi:10.1021/ic50177a056.
- ^ a b c d e f g h i j Keeler, James; Wothers, Peter (2014). Chemical Structure and Reactivity (2nd ed.). Oxford University Press. pp. 257–260. ISBN 978-0-19-9604135.
- ^ a b c d e f g h i j k l m n o p q r s t u v w x y Jensen, William B. (2000). «The Periodic Law and Table» (PDF). Archived from the original (PDF) on 10 November 2020. Retrieved 10 December 2022.
- ^ a b c Scerri, Eric (2009). «Which Elements Belong in Group 3?». Journal of Chemical Education. 86 (10): 1188. doi:10.1021/ed086p1188. Retrieved 1 January 2023.
- ^ a b Nefedov, V.I.; Trzhaskovskaya, M.B.; Yarzhemskii, V.G. (2006). «Electronic Configurations and the Periodic Table for Superheavy Elements» (PDF). Doklady Physical Chemistry. 408 (2): 149–151. doi:10.1134/S0012501606060029. ISSN 0012-5016. S2CID 95738861. Archived (PDF) from the original on 13 October 2016. Retrieved 15 August 2021.
- ^ a b Petrucci et al., pp. 326–7
- ^ Farberovich, O. V.; Kurganskii, S. I.; Domashevskaya, E. P. (1980). «Problems of the OPW Method. II. Calculation of the Band Structure of ZnS and CdS». Physica Status Solidi B. 97 (2): 631–640. Bibcode:1980PSSBR..97..631F. doi:10.1002/pssb.2220970230.
- ^ a b c d e Jensen, William B. (2015). «The positions of lanthanum (actinium) and lutetium (lawrencium) in the periodic table: an update». Foundations of Chemistry. 17: 23–31. doi:10.1007/s10698-015-9216-1. S2CID 98624395. Archived from the original on 30 January 2021. Retrieved 28 January 2021.
- ^ a b c d Hamilton, David C. (1965). «Position of Lanthanum in the Periodic Table». American Journal of Physics. 33 (8): 637–640. Bibcode:1965AmJPh..33..637H. doi:10.1119/1.1972042.
- ^ a b Jensen, W. B. (2015). «Some Comments on the Position of Lawrencium in the Periodic Table» (PDF). Archived from the original (PDF) on 23 December 2015. Retrieved 20 September 2015.
- ^ Wang, Fan; Le-Min, Li (2002). «镧系元素 4f 轨道在成键中的作用的理论研究» [Theoretical Study on the Role of Lanthanide 4f Orbitals in Bonding]. Acta Chimica Sinica (in Chinese). 62 (8): 1379–84.
- ^ Xu, Wei; Ji, Wen-Xin; Qiu, Yi-Xiang; Schwarz, W. H. Eugen; Wang, Shu-Guang (2013). «On structure and bonding of lanthanoid trifluorides LnF3 (Ln = La to Lu)». Physical Chemistry Chemical Physics. 2013 (15): 7839–47. Bibcode:2013PCCP…15.7839X. doi:10.1039/C3CP50717C. PMID 23598823.
- ^ Chi, Chaoxian; Pan, Sudip; Jin, Jiaye; Meng, Luyan; Luo, Mingbiao; Zhao, Lili; Zhou, Mingfei; Frenking, Gernot (2019). «Octacarbonyl Ion Complexes of Actinides [An(CO)8]+/− (An=Th, U) and the Role of f Orbitals in Metal–Ligand Bonding». Chem. Eur. J. 25 (50): 11772–11784. doi:10.1002/chem.201902625. PMC 6772027. PMID 31276242.
- ^ Singh, Prabhakar P. (1994). «Relativistic effects in mercury: Atom, clusters, and bulk». Physical Review B. 49 (7): 4954–4958. Bibcode:1994PhRvB..49.4954S. doi:10.1103/PhysRevB.49.4954. PMID 10011429.
- ^ Hu, Shu-Xian; Zou, Wenli (23 September 2021). «Stable copernicium hexafluoride (CnF6) with an oxidation state of VI+». Physical Chemistry Chemical Physics. 2022 (24): 321–325. Bibcode:2021PCCP…24..321H. doi:10.1039/D1CP04360A. PMID 34889909.
- ^ Seth, Michael; Schwerdtfeger, Peter; Fægri, Knut (1999). «The chemistry of superheavy elements. III. Theoretical studies on element 113 compounds». Journal of Chemical Physics. 111 (14): 6422–6433. Bibcode:1999JChPh.111.6422S. doi:10.1063/1.480168. S2CID 41854842.
- ^ Kelley, Morgan P.; Deblonde, Gauthier J.-P.; Su, Jing; Booth, Corwin H.; Abergel, Rebecca J.; Batista, Enrique R.; Yang, Ping (2018). «Bond Covalency and Oxidation State of Actinide Ions Complexed with Therapeutic Chelating Agent 3,4,3-LI(1,2-HOPO)». Inorganic Chemistry. 57 (9): 5352–5363. doi:10.1021/acs.inorgchem.8b00345. OSTI 1458511. PMID 29624372.
- ^ Johansson, B.; Abuja, R.; Eriksson, O.; et al. (1995). «Anomalous fcc crystal structure of thorium metal». Physical Review Letters. 75 (2): 280–283. Bibcode:1995PhRvL..75..280J. doi:10.1103/PhysRevLett.75.280. PMID 10059654.
- ^
Xu, Wen-Hua; Pyykkö, Pekka (8 June 2016). «Is the chemistry of lawrencium peculiar». Phys. Chem. Chem. Phys. 2016 (18): 17351–5. Bibcode:2016PCCP…1817351X. doi:10.1039/c6cp02706g. hdl:10138/224395. PMID 27314425. S2CID 31224634. Retrieved 24 April 2017. - ^ a b c Scerri, p. 354–6
- ^ Oganessian, Yu.Ts.; Abdullin, F.Sh.; Bailey, P.D.; Benker, D.E.; Bennett, M.E.; Dmitriev, S.N.; et al. (2010). «Synthesis of a new element with atomic number Z = 117«. Physical Review Letters. 104 (14): 142502. Bibcode:2010PhRvL.104n2502O. doi:10.1103/PhysRevLett.104.142502. PMID 20481935. S2CID 3263480. Archived from the original on 19 October 2021. Retrieved 15 August 2021.
- ^ Oganessian, Yu. T.; et al. (2002). «Results from the first 249
Cf+48
Ca experiment» (PDF). JINR Communication. Archived from the original (PDF) on 13 December 2004. Retrieved 13 June 2009. - ^ a b «IUPAC Announces the Names of the Elements 113, 115, 117, and 118». IUPAC. 30 November 2016. Archived from the original on 30 November 2016. Retrieved 1 December 2016.
- ^ National Institute of Standards and Technology (NIST) (August 2019). «Periodic Table of the Elements». Nist. Archived from the original on 8 February 2021. Retrieved 7 February 2021.
- ^ Fricke, B. (1975). Dunitz, J. D. (ed.). «Superheavy elements a prediction of their chemical and physical properties». Structure and Bonding. Berlin: Springer-Verlag. 21: 89–144. doi:10.1007/BFb0116496. ISBN 978-3-540-07109-9.
- ^ a b c d e f g h Connelly, N. G.; Damhus, T.; Hartshorn, R. M.; Hutton, A. T. (2005). Nomenclature of Inorganic Chemistry: IUPAC Recommendations 2005 (PDF). RSC Publishing. p. 51. ISBN 978-0-85404-438-2. Archived (PDF) from the original on 23 November 2018. Retrieved 26 November 2018.
- ^ a b c d e f g h i Fluck, E. (1988). «New Notations in the Periodic Table» (PDF). Pure Appl. Chem. 60 (3): 431–436. doi:10.1351/pac198860030431. S2CID 96704008. Archived (PDF) from the original on 25 March 2012. Retrieved 24 March 2012.
- ^ a b c d e f Scerri, Eric (18 January 2021). «Provisional Report on Discussions on Group 3 of the Periodic Table» (PDF). Chemistry International. 43 (1): 31–34. doi:10.1515/ci-2021-0115. S2CID 231694898. Archived (PDF) from the original on 13 April 2021. Retrieved 9 April 2021.
- ^ a b c d e f g h i William B. Jensen (1982). «The Positions of Lanthanum (Actinium) and Lutetium (Lawrencium) in the Periodic Table». J. Chem. Educ. 59 (8): 634–636. Bibcode:1982JChEd..59..634J. doi:10.1021/ed059p634.
- ^ a b c L. D. Landau, E. M. Lifshitz (1958). Quantum Mechanics: Non-Relativistic Theory. Vol. 3 (1st ed.). Pergamon Press. pp. 256–7.
- ^ a b Chemey, Alexander T.; Albrecht-Schmitt, Thomas E. (2019). «Evolution of the periodic table through the synthesis of new elements». Radiochimica Acta. 107 (9–11): 1–31. doi:10.1515/ract-2018-3082.
- ^ Pfeiffer, Paul (1920). «Die Befruchtung der Chemie durch die Röntgenstrahlenphysik». Naturwissenschaften (in German). 8 (50): 984–991. Bibcode:1920NW……8..984P. doi:10.1007/BF02448807. S2CID 7071495.
- ^ a b c d e f g h i Thyssen, P.; Binnemans, K. (2011). Gschneidner, K. A. Jr.; Bünzli, J-C.G; Vecharsky, Bünzli (eds.). Accommodation of the Rare Earths in the Periodic Table: A Historical Analysis. Handbook on the Physics and Chemistry of Rare Earths. Vol. 41. Amsterdam: Elsevier. pp. 1–94. doi:10.1016/B978-0-444-53590-0.00001-7. ISBN 978-0-444-53590-0.
- ^ Scerri, p. 375
- ^ a b «The constitution of group 3 of the periodic table». IUPAC. 2015. Archived from the original on 5 July 2016. Retrieved 30 July 2016.
- ^ a b c Lemonick, Sam (2019). «The periodic table is an icon. But chemists still can’t agree on how to arrange it». C&EN News. Archived from the original on 28 January 2021. Retrieved 16 December 2020.
- ^ a b Gray, p. 12
- ^ a b c d e Vlasov, L.; Trifonov, D. (1970). 107 Stories About Chemistry. Translated by Sobolev, D. Mir Publishers. pp. 23–27. ISBN 978-0-8285-5067-3.
- ^ a b Rayner-Canham, Geoffrey (2020). The Periodic Table: Past, Present, Future. World Scientific. pp. 53–70. ISBN 9789811218507.
- ^ Clayden, Jonathan; Greeves, Nick; Warren, Stuart; Wothers, Peter (2001). Organic Chemistry (1st ed.). Oxford University Press. ISBN 978-0-19-850346-0.
- ^ Seaborg, G. (1945). «The chemical and radioactive properties of the heavy elements». Chemical & Engineering News. 23 (23): 2190–93. doi:10.1021/cen-v023n023.p2190.
- ^ a b Kaesz, Herb; Atkins, Peter (2009). «A Central Position for Hydrogen in the Periodic Table». Chemistry International. 25 (6): 14. doi:10.1515/ci.2003.25.6.14.
- ^ a b Greenwood & Earnshaw, throughout the book
- ^ Scerri, Eric (2004). «The Placement of Hydrogen in the Periodic Table». Chemistry International. 26 (3): 21–22. doi:10.1515/ci.2004.26.3.21. Retrieved 1 January 2023.
- ^ Kurushkin, Mikhail (2020). «Helium’s placement in the Periodic Table from a crystal structure viewpoint». IUCrJ. 7 (4): 577–578. doi:10.1107/S2052252520007769. PMC 7340260. PMID 32695406. Archived from the original on 19 October 2021. Retrieved 19 June 2020.
- ^ a b Thyssen, Pieter; Ceulemans, Arnout (2017). Shattered Symmetry: Group Theory from the Eightfold Way to the Periodic Table. Oxford University Press. pp. 336, 360–381. ISBN 9780190611392.
- ^ a b c d Scerri, pp. 392−401
- ^ Grochala, Wojciech (1 November 2017). «On the position of helium and neon in the Periodic Table of Elements». Foundations of Chemistry. 20 (2018): 191–207. doi:10.1007/s10698-017-9302-7.
- ^ Bent Weberg, Libby (18 January 2019). ««The» periodic table». Chemical & Engineering News. 97 (3). Archived from the original on 1 February 2020. Retrieved 27 March 2020.
- ^ Grandinetti, Felice (23 April 2013). «Neon behind the signs». Nature Chemistry. 5 (2013): 438. Bibcode:2013NatCh…5..438G. doi:10.1038/nchem.1631. PMID 23609097.
- ^ a b c d e f Siekierski and Burgess, pp. 23–26
- ^ Siekierski and Burgess, p. 128
- ^ Lavelle, Laurence (2008). «Lanthanum (La) and Actinium (Ac) Should Remain in the d-block». Journal of Chemical Education. 85 (11): 1482–1483. Bibcode:2008JChEd..85.1482L. doi:10.1021/ed085p1482.
- ^ Johnson, David (1984). The Periodic Law (PDF). The Royal Society of Chemistry. ISBN 0-85186-428-7.
- ^ Wittig, Jörg (1973). «The pressure variable in solid state physics: What about 4f-band superconductors?». In H. J. Queisser (ed.). Festkörper Probleme: Plenary Lectures of the Divisions Semiconductor Physics, Surface Physics, Low Temperature Physics, High Polymers, Thermodynamics and Statistical Mechanics, of the German Physical Society, Münster, March 19–24, 1973. Advances in Solid State Physics. Vol. 13. Berlin, Heidelberg: Springer. pp. 375–396. doi:10.1007/BFb0108579. ISBN 978-3-528-08019-8.
- ^ a b c d e Greenwood and Earnshaw, pp. 27–9
- ^ Messler, R. W. (2010). The essence of materials for engineers. Sudbury, MA: Jones & Bartlett Publishers. p. 32. ISBN 978-0-7637-7833-0.
- ^ Myers, R. (2003). The basics of chemistry. Westport, CT: Greenwood Publishing Group. pp. 61–67. ISBN 978-0-313-31664-7.
- ^ Chang, R. (2002). Chemistry (7 ed.). New York: McGraw-Hill. pp. 289–310, 340–42. ISBN 978-0-07-112072-2.
- ^ Haas, Arthur Erich (1884-1941) Uber die elektrodynamische Bedeutung des Planckschen Strahlungsgesetzes und uber eine neue Bestimmung des elektrischen Elementarquantums und der dimension des wasserstoffatoms. Sitzungsberichte der kaiserlichen Akademie der Wissenschaften in Wien. 2a, 119 pp 119–144 (1910). Haas AE. Die Entwicklungsgeschichte des Satzes von der Erhaltung der Kraft. Habilitation Thesis, Vienna, 1909. Hermann, A. Arthur Erich Haas, Der erste Quantenansatz für das Atom. Stuttgart, 1965 [contains a reprint]
- ^ a b Clark, Jim (2012). «Atomic and Ionic Radius». Chemguide. Archived from the original on 14 November 2020. Retrieved 30 March 2021.
- ^ Cao, Chang-Su; Hu, Han-Shi; Li, Jun; Schwarz, W. H. Eugen (2019). «Physical origin of chemical periodicities in the system of elements». Pure and Applied Chemistry. 91 (12): 1969–1999. doi:10.1515/pac-2019-0901. S2CID 208868546. Retrieved 14 November 2022.
- ^ Kaupp, Martin (1 December 2006). «The role of radial nodes of atomic orbitals for chemical bonding and the periodic table». Journal of Computational Chemistry. 28 (1): 320–25. doi:10.1002/jcc.20522. PMID 17143872. S2CID 12677737.
- ^ a b c d Scerri, pp. 407–420
- ^ Greenwood and Earnshaw, p. 29
- ^ Kulsha, Andrey (2004). «Периодическая система химических элементов Д. И. Менделеева» [D. I. Mendeleev’s periodic system of the chemical elements] (PDF). primefan.ru (in Russian). Archived (PDF) from the original on 22 October 2020. Retrieved 17 May 2020.
- ^ Pyykkö, Pekka; Desclaux, Jean Paul (1979). «Relativity and the periodic system of elements». Accounts of Chemical Research. 12 (8): 276. doi:10.1021/ar50140a002.
- ^ Norrby, Lars J. (1991). «Why is mercury liquid? Or, why do relativistic effects not get into chemistry textbooks?». Journal of Chemical Education. 68 (2): 110. Bibcode:1991JChEd..68..110N. doi:10.1021/ed068p110.
- ^ Kulsha, A. V. «Есть ли граница у таблицы Менделеева?» [Is there a boundary to the Mendeleev table?] (PDF). www.primefan.ru (in Russian). Archived (PDF) from the original on 17 October 2020. Retrieved 8 September 2018.
- ^ Schädel, M. (2003). The Chemistry of Superheavy Elements. Dordrecht: Kluwer Academic Publishers. p. 277. ISBN 978-1-4020-1250-1.
- ^ Greenwood and Earnshaw, pp. 24–5
- ^ a b Clark, Jim (2016). «Ionisation Energy». Chemguide. Archived from the original on 22 April 2021. Retrieved 30 March 2021.
- ^ a b Clark, Jim (2012). «Electron Affinity». Chemguide. Archived from the original on 23 April 2021. Retrieved 30 March 2021.
- ^ Cárdenas, Carlos; Ayers, Paul; De Proft, Frank; Tozer, David J.; Geerlings, Paul (2010). «Should negative electron affinities be used for evaluating the chemical hardness?». Physical Chemistry Chemical Physics. 13 (6): 2285–2293. doi:10.1039/C0CP01785J. PMID 21113528.
- ^ Greenwood and Earnshaw, p. 113
- ^ a b c Siekierski and Burgess, pp. 45–54
- ^ a b c Siekierski and Burgess, pp. 134–137
- ^ a b Siekierski and Burgess, pp. 178–180
- ^ a b Scerri, pp. 14–15
- ^ a b c d Greenwood and Earnshaw, pp. 25–6
- ^ Allen, Leland C. (1989). «Electronegativity is the average one-electron energy of the valence-shell electrons in ground-state free atoms». Journal of the American Chemical Society. 111 (25): 9003–9014. doi:10.1021/ja00207a003.
- ^ Dieter, R. K.; Watson, R. T. (2009). «Transmetalation reactions producing organocopper compounds». In Rappoport, Z.; Marek, I. (eds.). The Chemistry of Organocopper Compounds. Vol. 1. John Wiley & Sons. pp. 443–526. ISBN 978-0-470-77296-6. Archived from the original on 17 October 2022. Retrieved 6 April 2022.
- ^ «Intermolecular bonding – van der Waals forces». Archived from the original on 22 January 2022. Retrieved 17 November 2021.
- ^ Clark, Jim (2019). «Metallic Bonding». Chemguide. Archived from the original on 21 April 2021. Retrieved 30 March 2021.
- ^ Pastor, G. M.; Stampfli, P.; Bennemann, K. (1988). «On the transition from Van der Waals- to metallic bonding in Hg-clusters as a function of cluster size». Physica Scripta. 38 (4): 623–626. Bibcode:1988PhyS…38..623P. doi:10.1088/0031-8949/38/4/022. S2CID 250842014.
- ^ a b c d e f g Siekierski and Burgess, pp. 60–66
- ^ McMinis, J.; Clay, R.C.; Lee, D.; Morales, M.A. (2015). «Molecular to Atomic Phase Transition in Hydrogen under High Pressure». Phys. Rev. Lett. 114 (10): 105305. Bibcode:2015PhRvL.114j5305M. doi:10.1103/PhysRevLett.114.105305. PMID 25815944.
- ^ Hawkes, Stephen J. (2001). «Semimetallicity?». Journal of Chemical Education. 78 (12): 1686. Bibcode:2001JChEd..78.1686H. doi:10.1021/ed078p1686.
- ^ Mewes, Jan-Michael; Smits, Odile Rosette; Jerabek, Paul; Schwerdtfeger, Peter (25 July 2019). «Oganesson is a Semiconductor: On the Relativistic Band-Gap Narrowing in the Heaviest Noble-Gas Solids». Angewandte Chemie. 58 (40): 14260–14264. doi:10.1002/anie.201908327. PMC 6790653. PMID 31343819.
- ^ Smith, J. D. (1973). The Chemistry of Arsenic, Antimony and Bismuth. Pergamon Press. p. 556.
- ^ a b Mewes, J.-M.; Smits, O. R.; Kresse, G.; Schwerdtfeger, P. (2019). «Copernicium is a Relativistic Noble Liquid». Angewandte Chemie International Edition. 58 (50): 17964–17968. doi:10.1002/anie.201906966. PMC 6916354. PMID 31596013.
- ^ a b Florez, Edison; Smits, Odile R.; Mewes, Jan-Michael; Jerabek, Paul; Schwerdtfeger, Peter (2022). «From the gas phase to the solid state: The chemical bonding in the superheavy element flerovium». The Journal of Chemical Physics. 157 (6): 064304. Bibcode:2022JChPh.157f4304F. doi:10.1063/5.0097642. PMID 35963734. S2CID 250539378.
- ^ a b
Gäggeler, H. W. (2007). «Gas Phase Chemistry of Superheavy Elements» (PDF). Paul Scherrer Institute. pp. 26–28. Archived from the original (PDF) on 20 February 2012. - ^ a b Ingo, Peter (15 September 2022). «Study shows flerovium is the most volatile metal in the periodic table». phys.org. Retrieved 22 November 2022.
- ^ a b Yakushev, A.; Lens, L.; Düllmann, Ch. E.; et al. (25 August 2022). «On the adsorption and reactivity of element 114, flerovium». Frontiers in Chemistry. 10 (976635): 976635. Bibcode:2022FrCh…10.6635Y. doi:10.3389/fchem.2022.976635. PMC 9453156. PMID 36092655.
- ^ Hermann, A.; Hoffmann, R.; Ashcroft, N. W. (2013). «Condensed Astatine: Monatomic and Metallic». Physical Review Letters. 111 (11): 116404-1–116404-5. Bibcode:2013PhRvL.111k6404H. doi:10.1103/PhysRevLett.111.116404. PMID 24074111.
- ^ Ball, Philip (13 September 2013). «Metallic properties predicted for astatine». Chemistry World. Retrieved 7 April 2023.
- ^ a b Clark, Jim (2012). «Metallic Structures». Chemguide. Archived from the original on 24 April 2021. Retrieved 30 March 2021.
- ^ Holleman, Arnold F.; Wiberg, Egon; Wiberg, Nils (1985). «Mangan». Lehrbuch der Anorganischen Chemie (in German) (91–100 ed.). Walter de Gruyter. pp. 1110–1117. ISBN 978-3-11-007511-3.
- ^ Wiberg, Egon; Wiberg, Nils & Holleman, Arnold Frederick (2001). Inorganic chemistry. Academic Press. p. 758. ISBN 978-0-12-352651-9.
- ^ Hammond, C. R. (2004). The Elements, in Handbook of Chemistry and Physics (81st ed.). Boca Raton (FL, US): CRC press. pp. 4–1. ISBN 978-0-8493-0485-9.
- ^ G.V. Samsonov, ed. (1968). «Mechanical Properties of the Elements». Handbook of the physicochemical properties of the elements. New York, USA: IFI-Plenum. pp. 387–446. doi:10.1007/978-1-4684-6066-7_7. ISBN 978-1-4684-6066-7. Archived from the original on 2 April 2015.
- ^ Hammer, B.; Norskov, J. K. (1995). «Why gold is the noblest of all the metals». Nature. 376 (6537): 238–240. Bibcode:1995Natur.376..238H. doi:10.1038/376238a0. S2CID 4334587.
- ^ Johnson, P. B.; Christy, R. W. (1972). «Optical Constants of the Noble Metals». Physical Review B. 6 (12): 4370–4379. Bibcode:1972PhRvB…6.4370J. doi:10.1103/PhysRevB.6.4370.
- ^ Clark, Jim (2018). «Atomic and Physical Properties of the Period 3 Elements». Chemguide. Archived from the original on 22 April 2021. Retrieved 30 March 2021.
- ^ a b Clark, Jim (2015). «The Trend From Non-Metal to Metal In the Group 4 Elements». Chemguide. Archived from the original on 27 April 2021. Retrieved 30 March 2021.
- ^ Wei, Lanhua; Kuo, P. K.; Thomas, R. L.; Anthony, T. R.; Banholzer, W. F. (1993). «Thermal conductivity of isotopically modified single crystal diamond». Physical Review Letters. 70 (24): 3764–3767. Bibcode:1993PhRvL..70.3764W. doi:10.1103/PhysRevLett.70.3764. PMID 10053956.
- ^ a b c d «Periodic Table of Chemical Elements». www.acs.org. American Chemical Society. 2021. Archived from the original on 3 February 2021. Retrieved 27 March 2021.
- ^ «Periodic Table». www.rsc.org. Royal Society of Chemistry. 2021. Archived from the original on 21 March 2021. Retrieved 27 March 2021.
- ^ a b c d e Seaborg, G. (c. 2006). «transuranium element (chemical element)». Encyclopædia Britannica. Archived from the original on 30 November 2010. Retrieved 16 March 2010.
- ^ Sherwin, E.; Weston, G. J. (1966). Spice, J. E. (ed.). Chemistry of the Non-Metallic Elements. Pergamon Press. ISBN 978-1-4831-3905-0.
- ^ a b Jensen, William B. (1986). «Classification, symmetry and the periodic table» (PDF). Comp. & Maths. With Appls. 12B (I/2). Archived (PDF) from the original on 31 January 2017. Retrieved 18 January 2017.
- ^ Greenwood and Earnshaw, pp. 29–31
- ^ Fernelius, W. C.; Loening, Kurt; Adams, Roy M. (1971). «Names of groups and elements». Journal of Chemical Education. 48 (11): 730–731. Bibcode:1971JChEd..48..730F. doi:10.1021/ed048p730.
- ^ Jensen, William B. (2003). «The Place of Zinc, Cadmium, and Mercury in the Periodic Table» (PDF). Journal of Chemical Education. 80 (8): 952–961. Bibcode:2003JChEd..80..952J. doi:10.1021/ed080p952. Archived from the original (PDF) on 11 June 2010. Retrieved 6 May 2012.
- ^ Leigh, G. J., ed. (2011). Principles of Chemical Nomenclature (PDF). The Royal Society of Chemistry. p. 9. ISBN 978-1-84973-007-5.
- ^ Winter, Mark (1993–2022). «WebElements». The University of Sheffield and WebElements Ltd, UK. Retrieved 5 December 2022.
- ^ Cowan, Robert D. (1981). The Theory of Atomic Structure and Spectra. University of California Press. p. 598. ISBN 9780520906150.
- ^ Villar, G. E. (1966). «A suggested modification to the periodic chart». Journal of Inorganic and Nuclear Chemistry. 28 (1): 25–29. doi:10.1016/0022-1902(66)80224-5.
- ^ Theuns, Tom. «Metallicity of stars». icc.dur.ac.uk. Durham University. Archived from the original on 27 September 2021. Retrieved 27 March 2021.
- ^ Burns, Gerald (1985). Solid State Physics. Academic Press, Inc. pp. 339–40. ISBN 978-0-12-146070-9.
- ^ Duffus, John H. (2002). ««Heavy Metals»–A Meaningless Term?» (PDF). Pure and Applied Chemistry. 74 (5): 793–807. doi:10.1351/pac200274050793. S2CID 46602106. Archived (PDF) from the original on 11 April 2021. Retrieved 27 March 2021.
- ^ Koppenol, W. (2016). «How to name new chemical elements» (PDF). Pure and Applied Chemistry. DeGruyter. doi:10.1515/pac-2015-0802. hdl:10045/55935. S2CID 102245448. Archived (PDF) from the original on 11 May 2020. Retrieved 15 August 2021.
- ^ Roth, Klaus (3 April 2018). «Is Element 118 a Noble Gas?». Chemie in unserer Zeit. doi:10.1002/chemv.201800029. Archived from the original on 2 March 2021. Retrieved 27 March 2021.
- ^ The Chemical Society of Japan (25 January 2018). «【お知らせ】高等学校化学で用いる用語に関する提案(1)への反応». www.chemistry.or.jp. The Chemical Society of Japan. Archived from the original on 16 May 2021. Retrieved 3 April 2021.
「12.アルカリ土類金属」の範囲についても,△を含めれば,すべての教科書で提案が考慮されている。歴史的には第4 周期のカルシウム以下を指していた用語だったが,「周期表の2 族に対応する用語とする」というIUPAC の勧告1)に従うのは現在では自然な流れだろう。
- ^ Wurzer, Ferdinand (1817). «Auszug eines Briefes vom Hofrath Wurzer, Prof. der Chemie zu Marburg» [Excerpt of a letter from Court Advisor Wurzer, Professor of Chemistry at Marburg]. Annalen der Physik (in German). 56 (7): 331–334. Bibcode:1817AnP….56..331.. doi:10.1002/andp.18170560709. Archived from the original on 8 October 2021. Retrieved 15 August 2021. Here, Döbereiner found that strontium’s properties were intermediate to those of calcium and barium.
- ^ Döbereiner, J. W. (1829). «Versuch zu einer Gruppirung der elementaren Stoffe nach ihrer Analogie» [An attempt to group elementary substances according to their analogies]. Annalen der Physik und Chemie. 2nd series (in German). 15 (2): 301–307. Bibcode:1829AnP….91..301D. doi:10.1002/andp.18290910217. Archived from the original on 8 October 2021. Retrieved 15 August 2021. For an English translation of this article, see: Johann Wolfgang Döbereiner: «An Attempt to Group Elementary Substances according to Their Analogies» (Lemoyne College (Syracuse, New York, USA)) Archived 9 March 2019 at the Wayback Machine
- ^ Horvitz, L. (2002). Eureka!: Scientific Breakthroughs That Changed The World. New York: John Wiley. p. 43. Bibcode:2001esbt.book…..H. ISBN 978-0-471-23341-1. OCLC 50766822.
- ^ Scerri, p. 47
- ^ Ball, P. (2002). The Ingredients: A Guided Tour of the Elements. Oxford: Oxford University Press. p. 100. ISBN 978-0-19-284100-1.
- ^ a b Chisholm, Hugh, ed. (1911). «Newlands, John Alexander Reina» . Encyclopædia Britannica. Vol. 19 (11th ed.). Cambridge University Press. p. 515.
- ^ Meyer, Julius Lothar; Die modernen Theorien der Chemie (1864); table on page 137 Archived 2 January 2019 at the Wayback Machine
- ^ Scerri, pp. 106–108
- ^ Scerri, p. 113
- ^ a b Scerri, pp. 117–123
- ^ Mendeleev, D. (1871). «The natural system of elements and its application to the indication of the properties of undiscovered elements». Journal of the Russian Chemical Society (in Russian). 3: 25–56. Archived from the original on 13 August 2017. Retrieved 23 August 2017.
- ^ Scerri, p. 149
- ^ Scerri, p. 151–2
- ^ Rouvray, R. «Dmitri Mendeleev». New Scientist. Archived from the original on 15 August 2021. Retrieved 19 April 2020.
- ^ Scerri, pp. 164–169
- ^ a b c Marshall, J.L.; Marshall, V.R. (2010). «Rediscovery of the Elements: Moseley and Atomic Numbers» (PDF). The Hexagon. Vol. 101, no. 3. Alpha Chi Sigma. pp. 42–47. S2CID 94398490. Archived from the original (PDF) on 16 July 2019. Retrieved 15 August 2021.
- ^ A. van der Broek, Physikalische Zeitschrift, 14, (1913), 32–41
- ^ Scerri, p. 185
- ^ A. van der Broek, Die Radioelemente, das periodische System und die Konstitution der Atom, Physik. Zeitsch., 14, 32, (1913).
- ^ E. Rutherford, Phil. Mag., 27, 488–499 (Mar. 1914). «This has led to an interesting suggestion by van Broek that the number of units of charge on the nucleus, and consequently the number of external electrons, may be equal to the number of the elements when arranged in order of increasing atomic weight. On this view, the nucleus charges of hydrogen, helium, and carbon are 1, 2, 6 respectively, and so on for the other elements, provided there is no gap due to a missing element. This view has been taken by Bohr in his theory of the constitution of simple atoms and molecules.»
- ^ Atkins, P. W. (1995). The Periodic Kingdom. HarperCollins Publishers, Inc. p. 87. ISBN 978-0-465-07265-1.
- ^ Egdell, Russell G.; Bruton, Elizabeth (2020). «Henry Moseley, X-ray spectroscopy and the periodic table». Philosophical Transactions of the Royal Society A: Mathematical, Physical and Engineering Sciences. 378 (2180). doi:10.1002/chem.202004775. PMID 32811359.
- ^ See Bohr table from 1913 paper below.
- ^ Helge Kragh, Aarhus, LARS VEGARD, ATOMIC STRUCTURE, AND THE PERIODIC SYSTEM, Bull. Hist. Chem., VOLUME 37, Number 1 (2012), p.43.
- ^ Scerri, pp. 208–218
- ^ Niels Bohr, «On the Constitution of Atoms and Molecules, Part III, Systems containing several nuclei» Philosophical Magazine 26:857—875 (1913)
- ^ Kragh, Helge (1 January 1979). «Niels Bohr’s Second Atomic Theory». Historical Studies in the Physical Sciences. 10: 123–186. doi:10.2307/27757389. ISSN 0073-2672. JSTOR 27757389.
- ^ W. Kossel, «Über Molekülbildung als Folge des Atom- baues,» Ann. Phys., 1916, 49, 229–362 (237).
- ^ Translated in Helge Kragh, Aarhus, LARS VEGARD, ATOMIC STRUCTURE, AND THE PERIODIC SYSTEM, Bull. Hist. Chem., VOLUME 37, Number 1 (2012), p.43.
- ^ Langmuir, Irving (June 1919). «The Arrangement of Electrons in Atoms and Molecules». Journal of the American Chemical Society. 41 (6): 868–934. doi:10.1021/ja02227a002. ISSN 0002-7863. Archived from the original on 26 January 2021. Retrieved 22 October 2021.
- ^ a b Bury, Charles R. (July 1921). «Langmuir’s Theory of the Arrangement of Electrons in Atoms and Molecules». Journal of the American Chemical Society. 43 (7): 1602–1609. doi:10.1021/ja01440a023. ISSN 0002-7863. Archived from the original on 30 October 2021. Retrieved 22 October 2021.
- ^ Jensen, William B. (2003). «The Place of Zinc, Cadmium, and Mercury in the Periodic Table» (PDF). Journal of Chemical Education. 80 (8): 952–961. Bibcode:2003JChEd..80..952J. doi:10.1021/ed080p952. Archived (PDF) from the original on 19 April 2012. Retrieved 18 September 2021.
The first use of the term «transition» in its modern electronic sense appears to be due to the British chemist C. R.Bury, who first used the term in his 1921 paper on the electronic structure of atoms and the periodic table
- ^ Scerri, pp. 218–23
- ^ Jensen, William B. (2007). «The Origin of the s, p, d, f Orbital Labels» (PDF). Journal of Chemical Education. 84 (5): 757–8. Bibcode:2007JChEd..84..757J. doi:10.1021/ed084p757. Archived from the original (PDF) on 23 November 2018. Retrieved 15 August 2021.
- ^ Karapetoff, Vladimir (1930). «A chart of consecutive sets of electronic orbits within atoms of chemical elements». Journal of the Franklin Institute. 210 (5): 609–624. doi:10.1016/S0016-0032(30)91131-3.
- ^ Ostrovsky, Valentin N. (2003). «Physical Explanation of the Periodic Table». Annals of the New York Academy of Sciences. 988 (1): 182–192. Bibcode:2003NYASA.988..182O. doi:10.1111/j.1749-6632.2003.tb06097.x. PMID 12796101. S2CID 21629328.
- ^ Klechkovskii, V.M. (1962). «Justification of the Rule for Successive Filling of (n+l) Groups». Journal of Experimental and Theoretical Physics. 14 (2): 334. Retrieved 23 June 2022.
- ^ a b Demkov, Yury N.; Ostrovsky, Valentin N. (1972). «n+l Filling Rule in the Periodic System and Focusing Potentials». Journal of Experimental and Theoretical Physics. 35 (1): 66–69. Bibcode:1972JETP…35…66D. Retrieved 25 November 2022.
- ^ Jørgensen, Christian Klixbüll (1988). «Influence of Rare Earths on Chemical Understanding and Classification». In Gschneidner Jr., Karl A.; Eyring, Leroy (eds.). Handbook on the Physics and Chemistry of Rare Earths. Vol. 11. Elsevier. pp. 197–292. ISBN 978-0-444-87080-3.
- ^ Kondō, Jun (January 1963). «Superconductivity in Transition Metals». Progress of Theoretical Physics. 29 (1): 1–9. Bibcode:1963PThPh..29….1K. doi:10.1143/PTP.29.1.
- ^ Scerri, pp. 313–321
- ^ Scerri, pp. 322–340
- ^ a b Seaborg, Glenn T. (1997). «Source of the Actinide Concept» (PDF). fas.org. Los Alamos National Laboratory. Archived (PDF) from the original on 15 August 2021. Retrieved 28 March 2021.
- ^ Scerri, pp. 356–9
- ^ Öhrström, Lars; Holden, Norman E. (2016). «The Three-letter Element Symbols». Chemistry International. 38 (2): 4–8. doi:10.1515/ci-2016-0204. S2CID 124737708. Archived from the original on 15 August 2021. Retrieved 28 March 2021.
- ^ Wapstra, A. H. (1991). «Criteria that must be satisfied for the discovery of a new chemical element to be recognized». Pure and Applied Chemistry. 63 (6): 879–886. doi:10.1351/pac199163060879. S2CID 95737691. Retrieved 18 October 2022.
- ^ «Names and symbols of transfermium elements (IUPAC Recommendations 1997)». Pure and Applied Chemistry. 69 (12): 2471–2474. 1997. doi:10.1351/pac199769122471.
- ^ Hofmann, Sigurd (2019). «Criteria for New Element Discovery». Chemistry International. 41 (1): 10–15. doi:10.1515/ci-2019-0103.
- ^ Scerri, E. (2012). «Mendeleev’s Periodic Table Is Finally Completed and What To Do about Group 3?». Chemistry International. 34 (4). doi:10.1515/ci.2012.34.4.28. Archived from the original on 5 July 2017.
- ^ Scerri, pp. 356–363
- ^ a b Chapman, Kit (30 November 2016). «What it takes to make a new element». Chemistry World. Royal Society of Chemistry. Archived from the original on 28 October 2017. Retrieved 22 March 2022.
{{cite journal}}: CS1 maint: bot: original URL status unknown (link)() - ^ Briggs, Helen (29 January 2019). «Happy birthday, periodic table». Archived from the original on 9 February 2019. Retrieved 8 February 2019.
- ^ Hofmann, Sigurd; Dmitriev, Sergey N.; Fahlander, Claes; Gates, Jacklyn M.; Roberto, James B.; Sakai, Hideyuki (4 August 2020). «On the discovery of new elements (IUPAC/IUPAP Report)». Pure and Applied Chemistry. 92 (9): 1387–1446. doi:10.1515/pac-2020-2926. S2CID 225377737.
- ^ a b Ball, P. (2019). «Extreme chemistry: experiments at the edge of the periodic table». Nature. 565 (7741): 552–555. Bibcode:2019Natur.565..552B. doi:10.1038/d41586-019-00285-9. ISSN 1476-4687. PMID 30700884.
- ^ Dmitriev, Sergey; Itkis, Mikhail; Oganessian, Yuri (2016). Status and perspectives of the Dubna superheavy element factory (PDF). Nobel Symposium NS160 – Chemistry and Physics of Heavy and Superheavy Elements. doi:10.1051/epjconf/201613108001. Archived (PDF) from the original on 28 August 2021. Retrieved 15 August 2021.
- ^ Sokolova, Svetlana; Popeko, Andrei (24 May 2021). «How are new chemical elements born?». jinr.ru. JINR. Archived from the original on 4 November 2021. Retrieved 4 November 2021.
- ^ Frazier, K. (1978). «Superheavy Elements». Science News. 113 (15): 236–38. doi:10.2307/3963006. JSTOR 3963006.
- ^ Fricke, B.; Greiner, W.; Waber, J. T. (1971). «The continuation of the periodic table up to Z = 172. The chemistry of superheavy elements». Theoretica Chimica Acta. 21 (3): 235–60. doi:10.1007/BF01172015. S2CID 117157377. Archived from the original on 19 October 2021. Retrieved 4 February 2020.
- ^ Pyykkö, P. (2011). «A suggested periodic table up to Z ≤ 172, based on Dirac–Fock calculations on atoms and ions». Physical Chemistry Chemical Physics. 13 (1): 161–68. Bibcode:2011PCCP…13..161P. doi:10.1039/c0cp01575j. PMID 20967377. S2CID 31590563. Archived from the original on 19 October 2021. Retrieved 11 January 2020.
- ^ a b Scerri, Eric (2020). «Recent attempts to change the periodic table». Philosophical Transactions of the Royal Society A. 378 (2180). Bibcode:2020RSPTA.37890300S. doi:10.1098/rsta.2019.0300. PMID 32811365. S2CID 221136189.
- ^ Jerabek, Paul; Schuetrumpf, Bastian; Schwerdtfeger, Peter; Nazarewicz, Witold (2018). «Electron and Nucleon Localization Functions of Oganesson: Approaching the Thomas-Fermi Limit». Phys. Rev. Lett. 120 (5): 053001. arXiv:1707.08710. Bibcode:2018PhRvL.120e3001J. doi:10.1103/PhysRevLett.120.053001. PMID 29481184. S2CID 3575243.
- ^ Schiff, L. I.; Snyder, H.; Weinberg, J. (1940). «On the Existence of Stationary States of the Mesotron Field». Physical Review. 57 (4): 315–318. Bibcode:1940PhRv…57..315S. doi:10.1103/PhysRev.57.315.
- ^ Reinhardt, Joachim; Greiner, Walter (2015). «Probing Supercritical Fields with Real and with Artificial Nuclei». Nuclear Physics: Present and Future. pp. 195–210. doi:10.1007/978-3-319-10199-6_19. ISBN 978-3-319-10198-9.
- ^ Pershina, Valeria (2020). «Relativistic effects on the electronic structure of the heaviest elements. Is the Periodic Table endless?» (PDF). Comptes Rendus Chimie. 23 (3): 255–265. doi:10.5802/crchim.25. S2CID 222225772. Archived (PDF) from the original on 11 December 2020. Retrieved 28 March 2021.
- ^ «Superheavy Element 114 Confirmed: A Stepping Stone to the Island of Stability». Berkeley Lab. 2009. Archived from the original on 20 July 2019. Retrieved 23 October 2019.
- ^ Oganessian, Yu. Ts. (2012). «Nuclei in the «Island of Stability» of Superheavy Elements». Journal of Physics: Conference Series. 337 (1): 012005. Bibcode:2012JPhCS.337a2005O. doi:10.1088/1742-6596/337/1/012005.
- ^ Schwerdtfeger, Peter; Pašteka, Lukáš F.; Punnett, Andrew; Bowman, Patrick O. (2015). «Relativistic and quantum electrodynamic effects in superheavy elements». Nuclear Physics A. 944 (December 2015): 551–577. Bibcode:2015NuPhA.944..551S. doi:10.1016/j.nuclphysa.2015.02.005.
- ^ Greiner, W. (2013). «Nuclei: superheavy-superneutronic-strange-and of antimatter» (PDF). Journal of Physics: Conference Series. 413 (1): 012002-1–012002-9. Bibcode:2013JPhCS.413a2002G. doi:10.1088/1742-6596/413/1/012002. Archived (PDF) from the original on 30 March 2019. Retrieved 15 August 2021.
- ^ Hofmann, Sigurd (2019). «Synthesis and properties of isotopes of the transactinides». Radiochimica Acta. 107 (9–11): 879–915. doi:10.1515/ract-2019-3104. S2CID 203848120.
- ^ Scerri, p. 386
- ^ Peter Möller (2016). «The limits of the nuclear chart set by fission and alpha decay | EPJ Web of Conferences». European Physical Journal Web of Conferences. Epj-conferences.org. 131: 03002. Bibcode:2016EPJWC.13103002M. doi:10.1051/epjconf/201613103002. Archived from the original on 20 June 2022. Retrieved 13 June 2022.
- ^ Holdom, B.; Ren, J.; Zhang, C. (2018). «Quark matter may not be strange». Physical Review Letters. 120 (1): 222001-1–222001-6. arXiv:1707.06610. Bibcode:2018PhRvL.120v2001H. doi:10.1103/PhysRevLett.120.222001. PMID 29906186. S2CID 49216916.
- ^ Cheng-Jun, Xia; She-Sheng, Xue; Ren-Xin, Xu; Shan-Gui, Zhou (2020). «Supercritically charged objects and electron-positron pair creation». Physical Review D. 101 (10): 103031. arXiv:2001.03531. Bibcode:2020PhRvD.101j3031X. doi:10.1103/PhysRevD.101.103031. S2CID 210157134.
- ^ Giuliani, S. A.; Matheson, Z.; Nazarewicz, W.; Olsen, E.; Reinhard, P.-G.; Sadhukhan, J.; Schtruempf, B.; Schunck, N.; Schwerdtfeger, P. (2019). «Colloquium: Superheavy elements: Oganesson and beyond». Reviews of Modern Physics. 91 (1): 011001-1–011001-25. Bibcode:2019RvMP…91a1001G. doi:10.1103/RevModPhys.91.011001. S2CID 126906074.
- ^ Giardina, G.; Fazio, G.; Mandaglio, G.; Manganaro, M.; Nasirov, A.K.; Romaniuk, M.V.; Saccà, C. (2010). «Expectations and limits to synthesize nuclei with Z ≥ 120». International Journal of Modern Physics E. 19 (5 & 6): 882–893. Bibcode:2010IJMPE..19..882G. doi:10.1142/S0218301310015333. Archived from the original on 19 October 2021. Retrieved 15 August 2021.
- ^ Zagrebaev, Valeriy; Karpov, Alexander; Greiner, Walter (2013). «Future of superheavy element research: Which nuclei could be synthesized within the next few years?» (PDF). Journal of Physics. IOP Publishing Ltd. 420 (1): 012001. arXiv:1207.5700. Bibcode:2013JPhCS.420a2001Z. doi:10.1088/1742-6596/420/1/012001. S2CID 55434734. Archived (PDF) from the original on 3 October 2015. Retrieved 1 December 2020.
- ^ Subramanian, S. (2019). «Making New Elements Doesn’t Pay. Just Ask This Berkeley Scientist». Bloomberg Businessweek. Archived from the original on 11 December 2019. Retrieved 18 January 2020.
- ^ Scerri, p. 20
- ^ Finding Aid to Edward G. Mazurs Collection of Periodic Systems Images. Science History Institute. Archived from the original on 27 March 2019. Retrieved 2 October 2018.
Click on ‘Finding Aid’ to go to full finding aid.
- ^ Francl, M. (May 2009). «Table manners» (PDF). Nature Chemistry. 1 (2): 97–98. Bibcode:2009NatCh…1…97F. doi:10.1038/nchem.183. PMID 21378810. Archived (PDF) from the original on 25 October 2012.
- ^ a b c Scerri, pp. 402–3
- ^ Scerri, Eric (29 January 2019). «Happy sesquicentennial to the periodic table of the elements». Oxford University Press. Archived from the original on 27 March 2019. Retrieved 12 April 2019.
- ^ Scerri, Eric (2021). «Various forms of the periodic table including the left-step table, the regularization of atomic number triads and first-member anomalies». ChemTexts. 8 (6). doi:10.1007/s40828-021-00157-8. S2CID 245540088.
Bibliography
- Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- Petrucci, Ralph H.; Harwood, William S.; Herring, F. Geoffrey (2002). General chemistry: principles and modern applications (8th ed.). Upper Saddle River, N.J: Prentice Hall. ISBN 978-0-13-014329-7. LCCN 2001032331. OCLC 46872308.
- Siekierski, S.; Burgess, J. (2002). Concise Chemistry of the Elements. Horwood. ISBN 978-1-898563-71-6.
- Scerri, Eric R (2020). The Periodic Table, Its Story and Its Significance, 2nd edition, Oxford University Press, New York, ISBN 978-0190914363.
Further reading
- Calvo, Miguel (2019). Construyendo la Tabla Periódica. Zaragoza, Spain: Prames. p. 407. ISBN 978-84-8321-908-9.
- Emsley, J. (2011). «The Periodic Table». Nature’s Building Blocks: An A–Z Guide to the Elements (New ed.). Oxford: Oxford University Press. pp. 634–651. ISBN 978-0-19-960563-7.
- Fontani, Marco; Costa, Mariagrazia; Orna, Mary Virginia (2007). The Lost Elements: The Periodic Table’s Shadow Side. Oxford: Oxford University Press. p. 508. ISBN 978-0-19-938334-4.
- Mazurs, E. G. (1974). Graphical Representations of the Periodic System During One Hundred Years. Alabama: University of Alabama Press. ISBN 978-0-19-960563-7.
- Rouvray, D.H.; King, R. B., eds. (2004). The Periodic Table: Into the 21st Century. Proceedings of the 2nd International Conference on the Periodic Table, part 1, Kananaskis Guest Ranch, Alberta, 14–20 July 2003. Baldock, Hertfordshire: Research Studies Press. ISBN 978-0-86380-292-8.
- Rouvray, D.H.; King, R. B., eds. (2006). The Mathematics of the Periodic Table. Proceedings of the 2nd International Conference on the Periodic Table, part 2, Kananaskis Guest Ranch, Alberta, 14–20 July 2003. New York: Nova Science. ISBN 978-1-59454-259-6.
- Scerri, E (n.d.). «Books on the Elements and the Periodic Table» (PDF). Archived (PDF) from the original on 11 August 2020. Retrieved 9 July 2018.
- Scerri, E.; Restrepo, G, eds. (2018). Mendeleev to Oganesson: A Multidisciplinary Perspective on the Periodic Table. Proceedings of the 3rd International Conference on the Periodic Table, Cuzco, Peru 14–16 August 2012. Oxford: Oxford University Press. ISBN 978-0-86380-292-8.
- van Spronsen, J. W. (1969). The Periodic System of Chemical Elements: A History of the First Hundred Years. Amsterdam: Elsevier. ISBN 978-0-444-40776-4.
- Verde, M., ed. (1971). Atti del convegno Mendeleeviano: Periodicità e simmetrie nella struttura elementare della materia [Proceedings of the Mendeleevian conference: Periodicity and symmetry in the elementary structure of matter]. 1st International Conference on the Periodic Table, Torino-Roma, 15–21 September 1969. Torino: Accademia delle Scienze di Torino.
External links
- Periodic Table featured topic page on Science History Institute Digital Collections featuring select visual representations of the periodic table of the elements, with an emphasis on alternative layouts including circular, cylindrical, pyramidal, spiral, and triangular forms.
- IUPAC Periodic Table of the Elements
- Dynamic periodic table, with interactive layouts
- Eric Scerri, leading philosopher of science specializing in the history and philosophy of the periodic table
- The Internet Database of Periodic Tables
- Periodic table of endangered elements
- Periodic table of samples
- Periodic table of videos
- WebElements
- The Periodic Graphics of Elements